A New Biomarker Risk Score May Improve Prognosis And Trials In Idiopathic Pulmonary Fibrosis
By Peter Schafer, Ph.D., chair, Prognostic Lung Fibrosis Consortium (PROLIFIC), Pulmonary Fibrosis Foundation
Idiopathic pulmonary fibrosis (IPF) is a progressive lung disease characterized by scarring of lung tissue that gradually impairs breathing and gas exchange. Despite decades of research, clinicians still lack approved prognostic biomarkers that can help predict how quickly the disease will progress in individual patients. Recently, an important step toward addressing this gap occurred when CDER accepted a letter of intent for the first IPF biomarker into its Biomarker Qualification Program. The biomarker tool, known as the Prognostic Lung Fibrosis Consortium (PROLIFIC) Risk Score, may help predict disease progression and improve clinical trial design.
The Need For Prognostic Biomarkers In IPF
For more than 20 years, scientists have investigated potential biomarkers for pulmonary fibrosis, yet none have been approved for use in clinical trials to stratify patients or determine eligibility. This lack of prognostic tools creates challenges for clinicians and researchers. IPF progresses unpredictably — some patients remain relatively stable for years, while others experience rapid declines in lung function or early mortality.
A reliable prognostic biomarker could help distinguish patients who are likely to remain stable from those at risk for rapid deterioration, acute exacerbations, or death. Such information would have important clinical implications. It could help physicians tailor treatment strategies, determine the optimal timing for therapies, and prioritize patients for limited resources such as lung transplantation.
For patients, improved prognostic insight could reduce uncertainty about the disease course and support informed decision-making. Individuals who learn they are at higher risk of rapid progression may be more motivated to begin or adhere to antifibrotic therapies, participate in clinical trials, or pursue earlier referral for lung transplantation.
To address this unmet need, industry and nonprofit partners, facilitated by the Pulmonary Fibrosis Foundation (PFF), established the PROLIFIC consortium. The consortium’s goal is to develop well-validated biomarker assays that can serve as prognostic or predictive tools in clinical trials involving patients with IPF.
Development Of The PROLIFIC Risk Score
First, the consortium surveyed the literature and identified 12 candidate blood protein biomarkers that had previously been reported to be associated with clinical outcomes in patients with IPF. Then it developed well-qualified assays to measure these 12 proteins in the serum. The biomarker tests were designed with strict quality controls to ensure precise and consistent measurements over time. This consistency is especially important in clinical trials, where patient enrollment often occurs gradually over one or two years. Reliable assays ensure that samples collected at different times produce comparable results. This robust assay development process may also help to gain acceptance by the regulatory authorities.
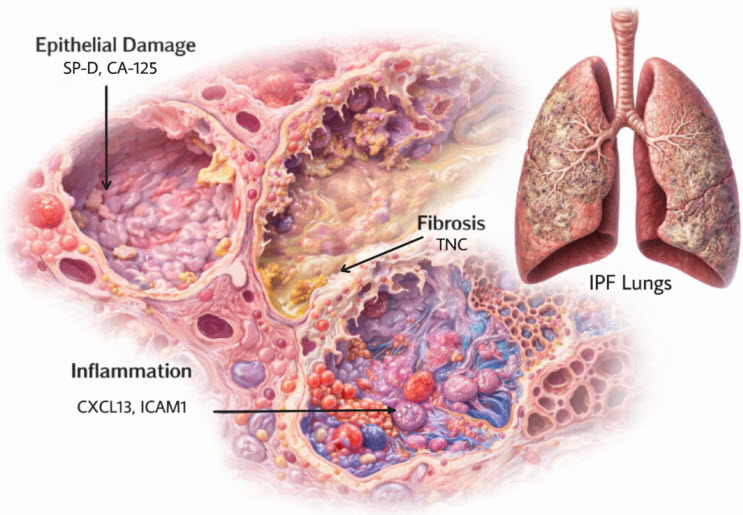
Figure 1. Disease processes occurring within the IPF lung and their representative biomarkers
Next, these candidate proteins were measured in the serum of patients collected from the Pulmonary Fibrosis Foundation Patient Registry, a large real-world database of patients with IPF and other interstitial lung diseases across the United States. Using statistical modeling, they identified the top biomarkers most strongly associated with clinical outcomes, specifically death, lung transplant, or a decline in lung function as measured by forced vital capacity (FVC). Five proteins were selected because of their prognostic associations with these outcomes, which collectively reflect key biological processes involved in IPF: lung cell injury, inflammation, and fibrosis or scarring (see Figure 1).
IPF typically begins when genetic and environmental factors trigger injury to epithelial cells in the lung, particularly alveolar type 2 (AT2) cells. These cells are essential for maintaining normal lung function and enabling efficient gas exchange. When AT2 cells are damaged or die, they release markers of epithelial injury into the bloodstream. Two such markers used in the PROLIFIC panel are surfactant protein D (SP-D) and cancer antigen 125 (CA-125).
In response to the epithelial damage, lung inflammation occurs. Immune cells such as macrophages and T cells are recruited to damaged lung tissue to address injury and potential infection. These immune cells release signaling proteins that contribute to inflammation. Two inflammatory markers included in the risk score are B lymphocyte chemoattractant (BLC, also known as CXCL13) and soluble intercellular adhesion molecule-1 (sICAM-1).
Following epithelial injury and inflammation, the lung attempts to repair itself. Fibroblasts begin producing collagen and other extracellular matrix proteins, resulting in fibrotic scarring that stiffens the lungs and makes breathing increasingly difficult. Tenascin C (TNC), another protein included in the panel, reflects this fibrotic process.
Predicting Disease Progression
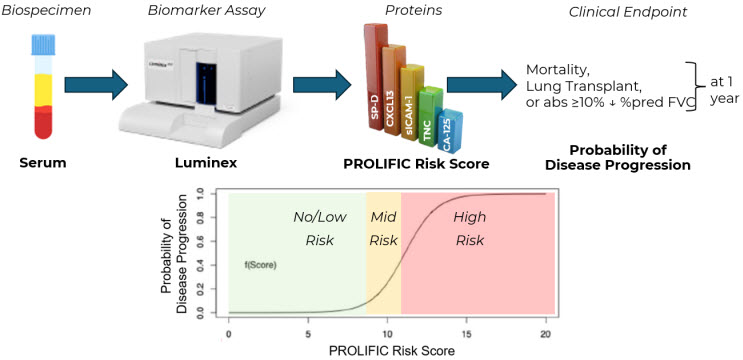
Figure 2. Measurement of IPF biomarkers in the serum and calculation of PROLIFIC Risk Score as a prognostic of disease progression.
The PROLIFIC Risk Score is calculated by measuring levels of the five selected proteins (SP-D, CA-125, CXCL13, sICAM-1, and TNC) in a patient’s serum (see Figure 2) using a quantitative multiplex assay with robust accuracy, precision, reproducibility, sensitivity, and specificity. The concentrations of these proteins are multiplied by weighting factors based on the strength of their association with the composite clinical outcome consisting of death, lung transplantation, or ≥10% absolute decline of % predicted FVC within one year. The PROLIFIC Risk Score is therefore prognostic, and it estimates the probability of the IPF patient experiencing any of these clinical outcomes.
Regulatory Progress And Next Steps
In April 2025, the PROLIFIC consortium submitted a letter of intent to the FDA’s Biomarker Qualification Program. In November 2025, the FDA accepted the submission, marking the first step toward official biomarker qualification. These documents are publicly available in the FDA archives under the Drug Development Tool Biomarker Qualification Program.
The next phase of the process involves providing additional evidence to demonstrate that the PROLIFIC Risk Score is a reliable prognostic tool for patients with IPF. Validation of the model is currently underway using data from the ISABELA1 and ISABELA2 randomized Phase 3 IPF clinical trials. Researchers are also validating the PROLIFIC Risk Score in samples from the PROFILE (Prospective Observation of Fibrosis in the Lung Clinical Endpoints) study in the United Kingdom. Further testing across multiple populations, such as rheumatoid arthritis-associated interstitial lung disease (RA-ILD) and other types of ILD will determine if the PROLIFIC Risk Score has utility beyond the IPF population, or if modifications to the score are needed for those more diverse patient groups.
If successful, the score could be used in clinical trials as a stratification factor or eligibility criterion. This would allow researchers to identify patients who are more likely to experience disease progression during a study, improving the efficiency and interpretability of trials.
The risk score is designed to categorize patients into low-risk and high-risk groups based on defined biomarker thresholds. This stratification could help ensure that clinical trials include patients most likely to experience measurable disease progression, thereby increasing the likelihood of detecting meaningful treatment effects.
Currently, the PROLIFIC biomarker assays are available for exploratory use in IPF clinical trials. Study sponsors are encouraged to incorporate the tool to further evaluate its performance and potential applications.
Potential Applications Beyond IPF
Although the PROLIFIC Risk Score was developed using data exclusively from patients with IPF, researchers are exploring whether the biomarker panel may also apply to other forms of pulmonary fibrosis. For example, studies are underway to examine its relevance in conditions such as rheumatoid arthritis–associated interstitial lung disease and other types of progressive pulmonary fibrosis.
If the biomarker panel proves useful across multiple fibrotic lung diseases, it could become an important tool for both clinical research and patient care.
Looking Ahead
The development of the PROLIFIC Risk Score represents a promising step toward improving prognostic assessment in IPF. By combining biomarkers that reflect epithelial injury, fibrosis, and inflammation, the tool offers a biologically informed method for predicting disease progression.
If successfully qualified by the FDA, the PROLIFIC Risk Score could significantly enhance clinical trial design, accelerate the development of new therapies, and ultimately provide clinicians and patients with clearer insight into the likely course of this challenging disease.
About The Author:
 Peter Schafer has 25 years of experience in drug discovery, research, and development in the pharmaceutical industry, and is now with the Pulmonary Fibrosis Foundation leading the PROLIFIC consortium. In his previous role as scientific vice president of translational medicine at Bristol Myers Squibb, Peter and his team designed and implemented biomarker plans for late-stage clinical trials in the areas of immunology, cardiovascular disease, and neuroscience. While at Celgene Corporation, he led the drug discovery biology group and helped to build the translational development group focused on hematology and immunology. He is co-inventor of the PDE4 inhibitor apremilast (Otezla®), now approved for psoriasis, psoriatic arthritis, and Behçet's Disease, for which he received the American Chemical Society Heroes of Chemistry Award in 2024. Peter obtained a Ph.D. in biochemistry, molecular biology, and cell biology from Northwestern University, and completed a post-doctoral research fellowship at the R. W. Johnson Pharmaceutical Research Institute.
Peter Schafer has 25 years of experience in drug discovery, research, and development in the pharmaceutical industry, and is now with the Pulmonary Fibrosis Foundation leading the PROLIFIC consortium. In his previous role as scientific vice president of translational medicine at Bristol Myers Squibb, Peter and his team designed and implemented biomarker plans for late-stage clinical trials in the areas of immunology, cardiovascular disease, and neuroscience. While at Celgene Corporation, he led the drug discovery biology group and helped to build the translational development group focused on hematology and immunology. He is co-inventor of the PDE4 inhibitor apremilast (Otezla®), now approved for psoriasis, psoriatic arthritis, and Behçet's Disease, for which he received the American Chemical Society Heroes of Chemistry Award in 2024. Peter obtained a Ph.D. in biochemistry, molecular biology, and cell biology from Northwestern University, and completed a post-doctoral research fellowship at the R. W. Johnson Pharmaceutical Research Institute.
