Clinical Trial Material Distribution Services
Source: Ropack Pharma Solutions
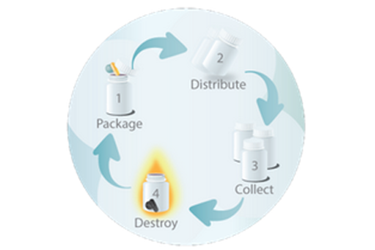
As your clinical material partner, we simplify – and accelerate – your clinical trials. From assistance with time-consuming international documentation through destruction following all regulatory protocols, RPS is your trusted clinical trial partner.
- Consolidated shipments that reduce costs
- Shortened go-to-market timelines
- Simplified regulatory documentation
- Canadian regulatory support and knowledge for processing
