Data Literacy: The Foundation For Modern Trial Execution
By Kristin Mauri
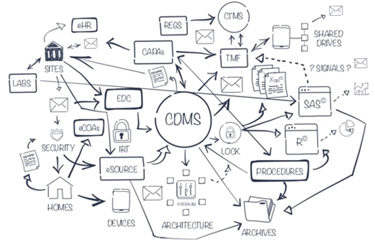
In 2016, the International Council for Harmonization (ICH) updated their “Guidelines for Good Clinical Practice.” One key shift was a mandate to implement a risk-based quality management system throughout all stages of a clinical trial, and to take a systematic, prioritized, risk-based approach to clinical trial monitoring—on-site monitoring, remote monitoring, or any combination thereof.
In theory, this new guidance freed researchers to take advantage of powerful new technologies that simplify remote monitoring and enable such monitoring to deliver study-wide insights that can speed answers and enhance patient safety. Yet in practice, clinical trial monitoring has remained grounded in on-site source data verification, a costly, time-consuming process that does not address risk or site performance data. It’s time for that to change. Data literacy is the change agent.
Get unlimited access to:
Enter your credentials below to log in. Not yet a member of Clinical Leader? Subscribe today.
