Decentralized Trials: Trends & Technology Propelling Home Healthcare Services
By Mathini Ilancheran, senior delivery lead - research, R&D, Beroe Inc.
The physical location of the study center is among the top three factors that hinder patients from participating in a clinical trial. Also, patient recruitment and retention have become challenges with the evolving number of diseases and trials. Moving some trial site activities into patients’ homes has been shown to increase recruitment by 60% or more, and it typically maintains patient retention at over 90%. As a result of COVID-19, the shift to home trials saw acceleration, with more digital tools capable of collecting clinically validated data. Pharma companies are outsourcing their home care services to third-party providers. However, some companies prefer to keep the management and strategic part in-house, such as training and education programs for nurses.
Home Healthcare Industry Trends
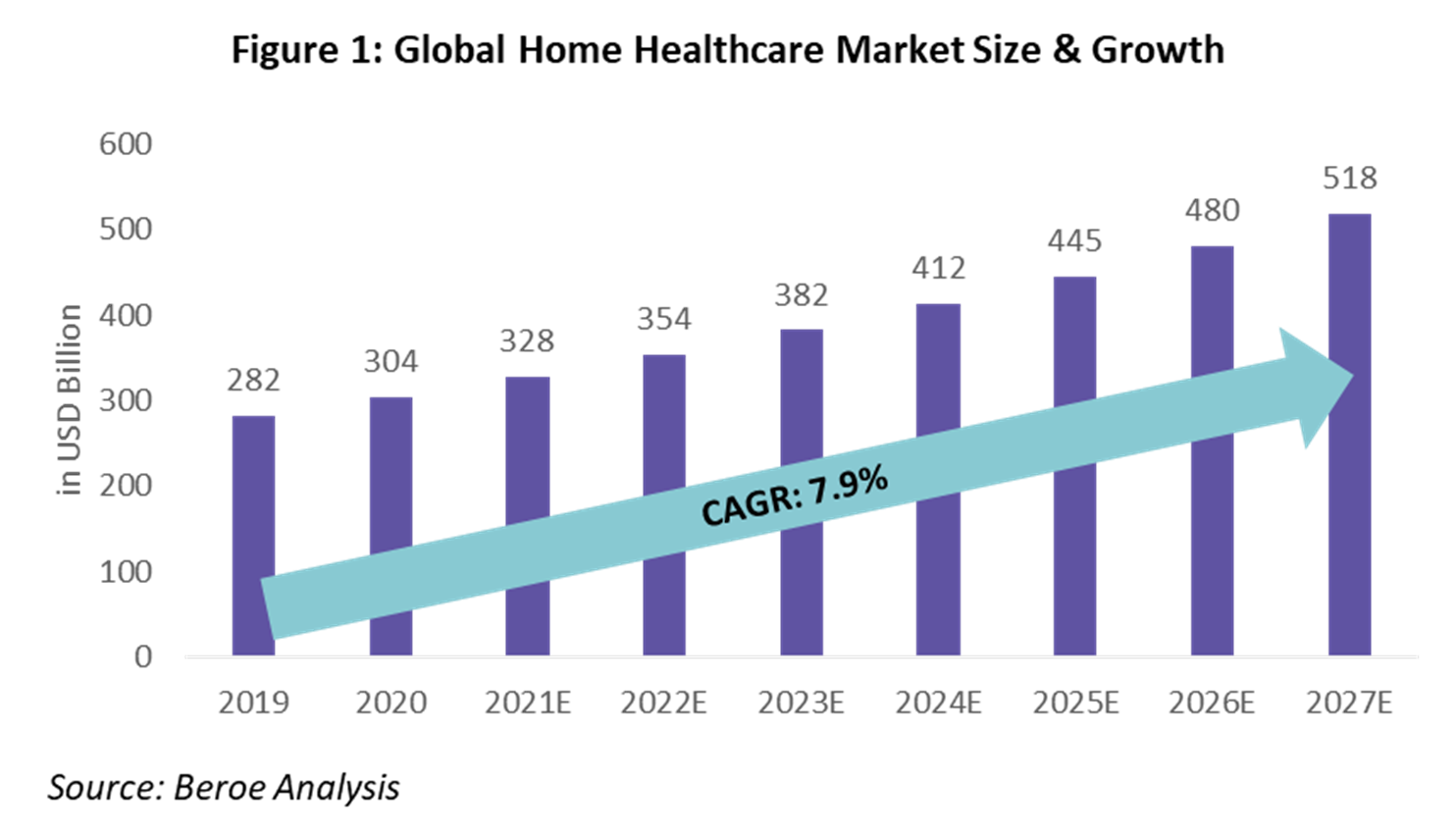
The global home healthcare market size is valued at approximately $328 billion in 2021 and is expected to grow at a rate of 7.9% over the period 2019 through 2027 (Figure 1). The focus by healthcare companies on improved patient outcomes and adherence to medication is the key driving factor, along with favorable regulations in the U.S. and European markets.
 Several key trends are evident within the home healthcare trial services industry. First, companies are entering into partnerships to acquire new technology. Collaborations that help in better technology transfer and in reaching rural areas have been a major trend with in-home visits. For example, insurance giant Humana is teaming up with DispatchHealth to provide access to hospital-level care in the home. The agreement will provide members living with multiple chronic conditions to be treated safely at home.
Several key trends are evident within the home healthcare trial services industry. First, companies are entering into partnerships to acquire new technology. Collaborations that help in better technology transfer and in reaching rural areas have been a major trend with in-home visits. For example, insurance giant Humana is teaming up with DispatchHealth to provide access to hospital-level care in the home. The agreement will provide members living with multiple chronic conditions to be treated safely at home.
We are seeing a greater focus on rare disease trials. Rare diseases have dispersed populations, making it difficult to reach the patients, which influences the sponsors to consider home healthcare services. This leads to a shift in focus toward hybrid models where sponsors identify aspects of a trial that could be done virtually or at locations closer to patients’ homes.
The shift to home-based care is leading to an expansion of facilities in some cases. Some global players in multiple regions are doubling their capacities to manage the increasing demand for logistics and also add the staff required for home-based care support. For example, Marken has expanded its Frankfurt cold chain facility to aid in quick turnaround times and faster delivery processes.
COVID-19 prompted a shift from care in nursing homes to healthcare services at home, resulting in an increased demand for skilled home healthcare nurses.
New FDA policies are encouraging companies to look at using home trials. Recent FDA guidance on conduct of clinical trials during COVID-19 advises pharma companies to evaluate alternative methods of patient safety assessments that can be deployed during times when travelling to sites may be restricted. In Europe, the central government has reimbursement benefits for this service, allowing people to choose the providers and type of healthcare.
Finally, the industry’s transition to new payment models is increasing adoption of home healthcare. A significant shift in payments from the fee for service model to value-based care is occurring. In this value-based model, the risk is high for service providers, allowing healthcare providers to experiment with new cost control models. Similar to cost-conscious consumers, providers are also turning to home healthcare solutions to save costs.
Use Of Technology In Home Healthcare Trials
According to Beroe Inc.’s interaction with experts, there has been an uptick in the use of automated patient access solutions between 2016 and 2020. More than 70% of trials use patient access solutions via hosted, web-based Software as a Service (SaaS) deployment methods for at least one function. The technologies most adopted by sponsors are:
- Artificial Intelligence: Using past patient data, patient demographics, chief complaints, location, and environmental factors, algorithms can predict who will show up late, who will cancel, and who will not show. Using the patient record, patient history, and physician habits, machine learning tools help set the right appointment length.
- Virtual Doctor Appointments: At-home tools for capturing clinical data allow doctors to extend the range of conditions that can be treated with virtual consultations. For example, MedWand is a device that lets doctors remotely measure the heart rate and oxygen levels and examine eyes, ears, and throat.
- Blockchain Technology: Blockchain securely communicates sensitive data among a network of parties such as researchers, doctors, and patients.
- mHealth: Mobile phone applications are being used to make healthcare more easily accessible. It can be an app reminding the user of a doctor’s appointment or when to take a pill, or a portable device that measures a person’s heart rate on the go. It implies real-time access that supports patient engagement.
- Telehealth: Telehealth makes it possible for doctors to reach a larger number of patients and can help manage chronic conditions without having to visit the doctor every time the patient encounters a trivial problem.
- Interactive Voice Response (IVR): Communication via text or voicemail provides a way for health groups to reduce no-shows, as well as eliminate outstanding balances.
- Patient Portals: An online portal is a place where patients go to confirm appointments, fill out their medical and social history forms, update health records, view lab results, and pay any outstanding balances.
- Kiosks: These have great potential for collecting patient health information, delivering access to healthcare information, and providing patient education. These can be used in both clinic and hospital settings.
- Call Centers: Hospitals and other medical practices use third-party organizations to call patients prior to their appointment time to collect the data required for their visit. Medical history data is collected by a call center agent and entered directly into the care facility's registration system and EHR for storage and later use.
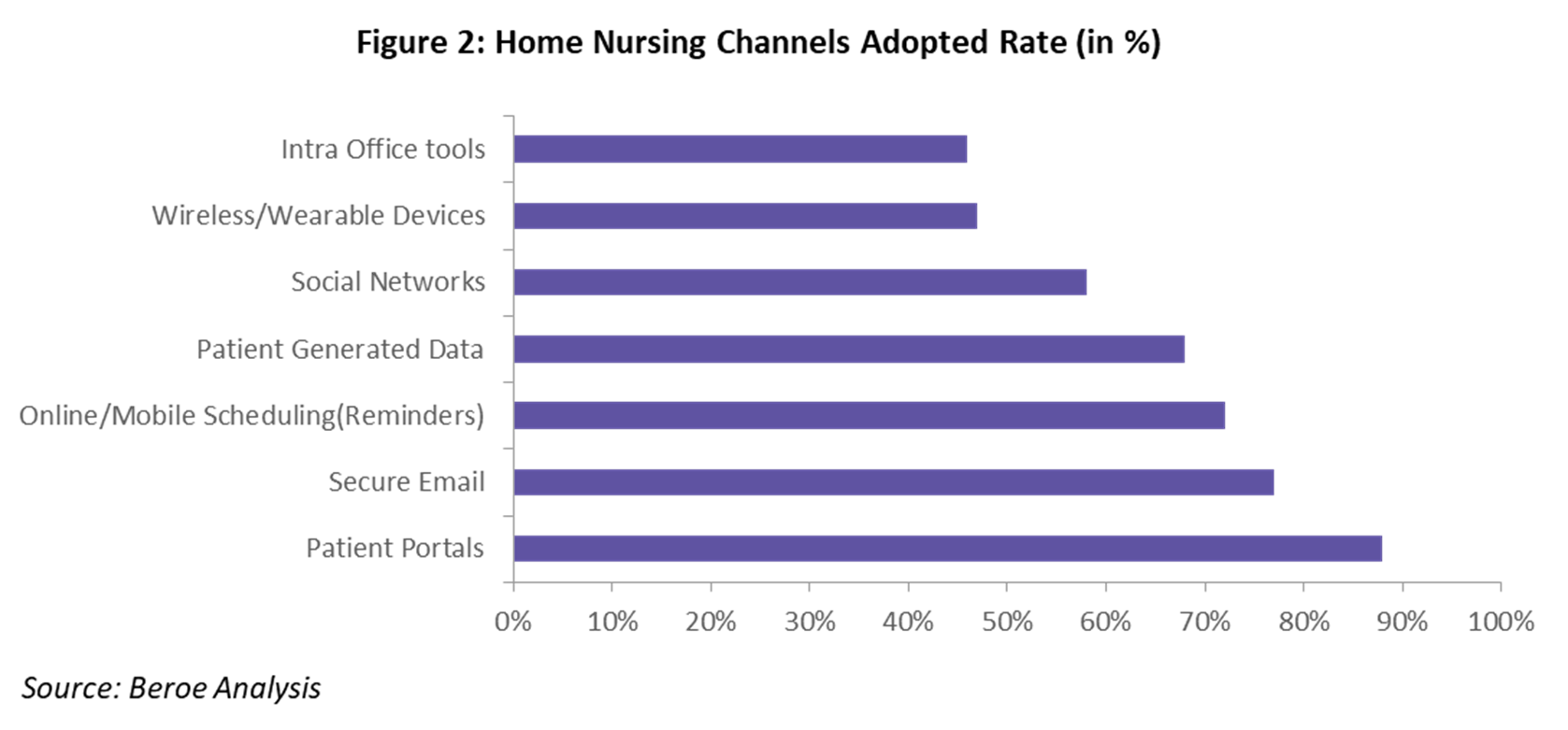
A variety of communication channels are used in home nursing services. Technologies for virtual communities include online message boards, automatic mailing list servers for asynchronous communication, videoconferencing, internet relay chat, group and private chat rooms for synchronous communication, and social networking platforms, such as Facebook or Twitter. Figure 2 provides the adoption percentage for each different channel.
The Future Of Home Healthcare Services
Home care services rely on digital tools and engagement that can help treat patients across the globe and also increase patient access. Apart from digitization, there is a significant increase in demand for home care nurses and portable home devices that generate data to a centralized system, aiding in better data monitoring. The pandemic has forced sponsors to shift their focus to decentralized trials and virtual engagement so patients can afford to receive their treatment at home. This is possible with a global network of distributors experienced with domestic and international logistics, strictly adhering to good manufacturing practices (GMP) and good distribution practices (GDP), and with extensive experience in clinical trial space. With a higher level of protocol management and supply chain complexity involved, at-home kits require the coordination of various systems and stakeholders to reach patients at the right time at their preferred location. Eventually, the adoption of at-home care will provide an opportunity to launch promising therapies in the future.
About The Author:
 Mathini Ilancheran is the principal analyst of R&D for Beroe Inc. She specializes in understanding market scenarios across the globe in the outsourcing arena. Her analysis has enabled global fortune 500+ companies in their strategic decisions on supply base optimization, risk reduction, innovation and efficient sourcing. She has written for several publications related to R&D procurement opportunities. With her category knowledge, she has published 35+ articles in leading journals, co-authored with industry experts. She has a master's in management from University College London (UCL) and has worked as a consultant in the U.K. You can contact her at mathini.ilancheran@beroe-inc.com and connect with her on LinkedIn.
Mathini Ilancheran is the principal analyst of R&D for Beroe Inc. She specializes in understanding market scenarios across the globe in the outsourcing arena. Her analysis has enabled global fortune 500+ companies in their strategic decisions on supply base optimization, risk reduction, innovation and efficient sourcing. She has written for several publications related to R&D procurement opportunities. With her category knowledge, she has published 35+ articles in leading journals, co-authored with industry experts. She has a master's in management from University College London (UCL) and has worked as a consultant in the U.K. You can contact her at mathini.ilancheran@beroe-inc.com and connect with her on LinkedIn.

