How Important Are Medical Monitors Under ICH E6(R3)?
By Dora V. Ardila, MBBS, Ph.D., MBA candidate, Healthcare Consultant

As ICH E6(R3) guides researchers toward proactive, risk-proportionate quality management,1,2 the clinical research physician, or medical monitor (MM), plays an increasingly critical and often underrecognized role in ensuring patient safety, regulatory compliance, and scientific integrity.3,4 Under E6(R3), their responsibilities extend beyond traditional adverse event (AE) adjudication to encompass quality by design (QbD) protocol development, critical-to-quality (CtQ) factor identification, and risk-proportionate oversight that protects both patients and data integrity.5
In particular, the integration of physician-led medical governance determines whether fast start-up translates into sustainable quality or downstream regulatory vulnerability.6 This article demonstrates why medical monitor-led implementation of E6(R3) outperforms operations-only models across six critical performance dimensions.
The Evolution Of Medical Monitoring Under ICH E6(R3)
From Reactive Compliance to Proactive Risk Governance
The progression from ICH E6(R2) to E6(R3) reflects a maturation in regulatory philosophy. Where E6 (R2) introduced risk-based monitoring concepts, E6(R3) operationalizes comprehensive risk-based quality management (RBQM) that integrates proactive risk assessment at all trial stages.7,8 The guideline emphasizes QbD principles, reducing unnecessary documentation while focusing resources on factors critical to participant protection and data reliability.9
E6(R3) explicitly recognizes that quality cannot be inspected into trials retrospectively — it must be engineered upstream through medically informed protocol design, monitoring strategies, and oversight frameworks.10 This paradigm shift elevates the medical monitor from a consultative safety advisor to a core governance function with accountability spanning protocol development through database lock.
Defining The Medical Monitor Role
The medical monitor serves as a dedicated physician and clinical scientist responsible for ongoing safety oversight and trial integrity across all phases of clinical development.3,11 Core responsibilities include:
- protocol review and CtQ factor identification
- pre-enrollment eligibility assessment in complex studies
- real-time safety surveillance and AE causality determination
- medical guidance to investigative sites and study teams
- benefit-risk reassessment throughout trial conduct
- unblinding decision-making for severe safety events
- regulatory communication and inspection preparedness
- strategic go/no-go recommendations based on integrated data synthesis.
In early-phase oncology trials, where extensive monitoring of adverse events is essential given the toxicological characteristics of anti-cancer agents, the medical monitor’s role becomes particularly critical.12 The medical monitor evaluates procedural burden, identifies unnecessary or duplicative assessments, and ensures comprehensive safety monitoring plans are embedded in protocols.3
Evidence-Based Performance Advantages Of Medical Monitor-Led E6(R3) Implementation
Protocol Design: Quality by Design Vs. Operational Reactivity
Proposed Scenario: Operations-Focused Approach
The following is based on personal experience with a Phase 2 oncology trial launched under Australia's CTN pathway. The feasibility assessment focused predominantly on-site capacity and recruitment projections without structured medical input. The protocol contained overly restrictive inclusion criteria, ambiguous safety monitoring windows, and biomarker endpoints lacking clear CtQ definitions.
Results within nine months:
- Two protocol amendments required
- Three-month recruitment pause
- Estimated cost per amendment: AUD $350,000 to $500,000
- Investor confidence impacted
Proposed Scenario: Medical Monitor-Led E6(R3) Implementation
Again, this, too, is based on personal experience. Before regulatory submission, the medical monitor conducted a structured CtQ mapping aligned with E6(R3) principles. The inclusion criteria were optimized for recruitment feasibility while maintaining scientific rigor. Biomarker endpoints were aligned with operational capabilities and regulatory expectations. Finally, proactive safety trigger thresholds were defined with explicit escalation pathways.
Results:
- Zero major amendments
- Recruitment variance reduced
- Site queries reduced
- Database lock accelerated by at least Six weeks
Key Insight: Protocol amendments represent one of the most expensive and timeline-disruptive events in clinical development. Medical clarity upstream prevents operational correction downstream.
Safety Governance: Reactive Reporting Vs. Signal Intelligence
Traditional safety monitoring emphasizes procedural compliance — SAE processing timelines, query resolution metrics, and periodic safety committee meetings. This reactive approach often detects safety signals only after regulatory notification thresholds are crossed.13
Continuous medical and safety monitoring during clinical trials enables earlier detection of safety concerns through systematic data review and trend analysis.14,15 Interactive visualization tools facilitate efficient detection of data inconsistencies and enable more effective medical oversight.14
Medical Monitor-Led Proportionate Risk Surveillance Under E6(R3)
Implementing E6(R3) principles, the medical monitor establishes:
- predefined high-risk AE clusters based on mechanism of action
- aggregated safety trend dashboards reviewed every two weeks
- AI-assisted anomaly detection for subclinical parameter shifts
- predefined dose modification rules and stopping criteria.
In one documented implementation, subclinical signals were detected in laboratory parameter trends at Week 8, enabling dosing protocol adjustment before regulatory escalation became necessary.6 This proactive intervention avoided site suspension, maintained enrollment velocity, and preserved sponsor credibility — delivering signal detection four to six weeks earlier than conventional event-based monitoring.
Quantitative Safety Monitoring
Bayesian statistical frameworks provide analytical flexibility for safety surveillance by incorporating existing knowledge into decision-making and enabling integration of data from multiple sources.16 These methods support both blinded and unblinded monitoring strategies, allowing sponsors to establish up-front safety criteria for serious adverse events and adverse events of special interest (AESIs).16
Data Integrity: Volume Vs. Decision-Relevant Data
E6(R3)'s emphasis on CtQ factors challenges the conventional assumption that comprehensive data collection ensures trial quality.17 In reality, excessive data collection without medical prioritization generates query noise, delays adjudication, and obscures critical safety signals.
Operations-Centric Monitoring
Large volumes of data collected from ePRO, wearables, imaging, and laboratory panels without CtQ prioritization result in:
- increased query backlogs
- endpoint adjudication delays
- increased statistical variability
- resource misallocation to non-critical data points.
Medical Monitor-Led CtQ Focus Under R3
The medical monitor identifies three endpoints critical to regulatory success, two safety parameters requiring enhanced review, and categorizes digital metrics as exploratory rather than core. Monitoring intensity is reallocated accordingly, resulting in:
- reduction in non-critical queries
- accelerated interim analysis readiness
- lower monitoring costs.
Key Insight: More data does not equal better decisions. E6(R3) requires medically-informed prioritization to focus resources on factors truly critical to participant protection and data reliability.
Vendor Oversight: Delegation Does Not Dilute Accountability
E6(R3) explicitly reinforces that outsourcing clinical trial activities does not dilute sponsor accountability.18,19 Sponsors must maintain active oversight through structured risk assessment, centralized monitoring, and traceable decision-making throughout the study life cycle.19.
Operations-Only CRO Oversight Model
When medical oversight is limited to periodic review, risks emerge:
- Inconsistent causality narratives across sites
- Fragmented benefit-risk documentation
- Inspection vulnerability due to missing medical rationale
Medical Monitor-Governed Oversight Model
The medical monitor actively participates in:
- medical review of monitoring plans and CtQ alignment
- governance meeting participation with documented decisions
- benefit-risk reassessments at predefined milestones
- validation of escalation triggers and protocol deviation assessments.
Outcomes include reduced audit findings, continuous inspection-ready documentation, and stronger cross-functional clarity between operational execution and medical accountability.
Key Insight: Operations optimize workflow efficiency. Medical monitors optimize regulatory defensibility and scientific integrity.
Portfolio-Level Capital Efficiency
In resource-constrained early-stage biotechnology companies, capital preservation determines survival. The medical monitor synthesizes recruitment velocity, emerging safety trends, interim efficacy signals, and operational burden metrics to provide structured go/no-go recommendations.6
This strategic medical decision-making transcends operational project management, directly impacting investor confidence and runway optimization.
Comparative Performance Framework
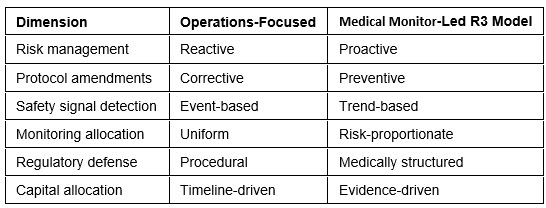
Table 1: Performance comparison of operations-focused versus medical monitor-led E6(R3) implementation models
Quality Frameworks For Patient Protection Under E6(R3)
Core Quality Principles
E6(R3) articulates 13 fundamental principles, with Principle 6 specifically addressing quality management systems.20 The quality framework must ensure:
- Risk-based quality management (RBQM): Systematic identification of factors critical to quality with proportionate oversight allocation7,21
- Quality by design: Proactive integration of quality considerations from protocol development through closeout9,21
- Critical-to-quality factor emphasis: Concentration of resources on elements essential to participant protection and data reliability5,21
- Traceable decision-making: Documentation of risk assessments, benefit-risk evaluations, and protocol modifications with clear medical rationale19
- Proportionate monitoring: Tailored oversight intensity based on study complexity, phase, therapeutic area, and identified risks22
Patient Rights and Safety Protection
The ethical foundation of clinical research requires protection of participant rights, safety, and well-being.23 E6(R3) emphasizes:
- informed consent processes that accurately communicate risks and benefits
- independent ethics committee oversight aligned with contemporary trial methodologies
- safety monitoring plans with predefined intervention thresholds
- data monitoring committees (DMCs) for trials with significant safety uncertainty16
- transparent adverse event reporting to participants, investigators, and regulators
- protection of vulnerable populations through enhanced safeguards.
Institutional Quality Management Systems
Organizations conducting clinical trials must implement comprehensive quality management systems encompassing20:
- documented standard operating procedures (SOPs) aligned with E6(R3)
- ongoing training programs for all trial personnel
- risk-based monitoring plans with medical input
- regular quality audits focusing on CtQ factors
- corrective and preventive action (CAPA) systems
- document management ensuring traceability and inspection readiness.
The shift toward QbD requires that sponsors establish cross-functional oversight meeting rhythms with traceable actions rather than status updates.19 This proactive governance model embeds medical expertise throughout trial conduct rather than relegating it to reactive consultation.
Implementation Road Map: Embedding Medical Monitor-Led Governance Under E6(R3)
Protocol Development Phase
- Engage the medical monitor early in protocol conceptualization.
- Conduct joint CtQ mapping sessions with ClinOps, medical affairs, data management, and quality assurance.
- Define medically appropriate eligibility criteria, balancing scientific rigor with recruitment feasibility.
- Establish proactive safety monitoring thresholds and escalation triggers.
- Design risk-proportionate monitoring strategies focusing resources on CtQ factors.
Trial Initiation Phase
- Medical monitor participates in investigator meetings to provide medical context and safety guidance.
- Conduct a medical review of site qualification from a clinical capability perspective.
- Establish safety surveillance dashboards with trend visualization.
- Define medical monitor communication pathways for real-time site queries.
- Integrate with DSMB structures where applicable.
Ongoing Conduct Phase
- Hold regular safety surveillance meetings with structured benefit-risk reassessments.
- Aggregate data review at predefined intervals (typically, every two weeks).
- Document medical decision-making for protocol deviations and amendments.
- Provide continuous medical input into monitoring visit findings and CAPA plans.
- Hold periodic governance meetings with traceable decisions and rationales.
Study Closeout and Regulatory Readiness
- Perform a medical review of final study reports emphasizing a benefit-risk narrative.
- Integrate safety summaries with causality assessments.
- Prepare inspection-ready medical documentation.
- Integrate lessons learned into subsequent protocol development.
Future Directions: Technology Integration and Medical Oversight
The E6(R3) framework explicitly encourages technology adoption to enhance quality, efficiency, and participant protection.2,9 Emerging technologies augmenting medical monitor capabilities include:
- AI-assisted safety surveillance: Machine learning algorithms for early anomaly detection in laboratory parameters, vital signs, and AE patterns14
- Data visualization platforms: Interactive dashboards enabling rapid pattern recognition across sites and cohorts32
- Centralized monitoring systems: Real-time data review reducing site visit burden while enhancing oversight quality33
- EHR integration: Streamlined source data verification and reduced documentation burden
- Decentralized trial technologies: Remote patient monitoring requiring adapted medical oversight frameworks34
These innovations do not replace physician judgment — they amplify it, enabling medical monitors to synthesize larger data sets more efficiently and detect subtle signals earlier in trial conduct.
Regulatory Landscape: Global Harmonization And Regional Implementation
The adoption of ICH GCP E6(R3) by the FDA,2 EMA,35 and Australia's TGA represents a harmonized expectation for risk-based quality management globally. However, regional nuances persist:
- FDA: Emphasis is on critical thinking, proportionality, and flexibility supporting diverse trial designs while maintaining participant protection and result reliability.2
- EMA: Focus is on quality culture, proactive quality design, and stakeholder engagement using risk-based approaches.35
- TGA: Aligns with ICH standards while maintaining Australia's streamlined CTN pathway and R&D incentive structures.
Sponsors conducting multinational development programs must ensure their medical governance frameworks satisfy the most stringent requirements across jurisdictions.
Conclusion
ICH E6(R3) transforms the medical monitor from a reactive safety consultant into a strategic performance multiplier. When embedded correctly, medical monitor-led governance delivers measurable improvements across six critical dimensions: protocol quality, safety surveillance, data integrity, monitoring efficiency, regulatory defensibility, and capital efficiency. The distinction between operational execution and medical governance is financial, regulatory, and strategic. Focusing exclusively on clinical operations improves logistics; embedding medical monitor-led E6(R3) governance improves outcomes.
The question facing sponsors is no longer whether to implement risk-based medical governance under E6(R3), but how rapidly and comprehensively they can transform organizational culture to embed medical expertise at the center of quality management systems. Those who make this transition decisively will define the next generation of clinical development excellence.
References:
- U.S. Food and Drug Administration. (2025). E6(R3) Good Clinical Practice (GCP) - Guidance for Industry. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/e6r3-good-clinical-practice-gcp
- Sidley Austin LLP. (2025). U.S. FDA's Adoption of ICH E6(R3) Good Clinical Practice: Key Takeaways for Sponsors. https://www.sidley.com/en/insights/newsupdates/2025/12/
- Byrth, F., et al. (2025). The Essential Role of Medical Monitors in Clinical Trials. Journal of Clinical Research Best Practices, 21(3), 1-12. https://pmc.ncbi.nlm.nih.gov/articles/PMC12527326/
- Clinical Leader. (2022). How To Make Trials More Successful With Medical Monitors. https://www.clinicalleader.com/doc/how-to-make-trials-more-successful-with-medical-monitors-0001
- Advarra. (2025). The Evolution of Clinical Quality: Key Considerations from ICH E6 R3. https://www.advarra.com/blog/the-evolution-of-clinical-quality-key-considerations-from-ich-e6-r3/
- The Clinical Research Physician Under ICH GCP E6(R3): Why Risk-Based Medical Governance Outperforms Operations-Centric Trial Models. (2026). White Paper. [Internal document]
- Cyntegrity. (2026). ICH E6(R3) Sponsor Responsibilities Explained. https://cyntegrity.com/ich-e6-r3-sponsor-responsibilities/
- Patel, S., & Kumar, R. (2023). The revamped Good Clinical Practice E6(R3) guideline: Key amendments and their implications. Perspectives in Clinical Research, 14(4), 151-155. https://pmc.ncbi.nlm.nih.gov/articles/PMC10679570/
- Singh, A., et al. (2025). International Council for Harmonisation E6 (R3): The Good Clinical Practice Guideline Evolution. Perspectives in Clinical Research, 16(2), 45-52. https://pmc.ncbi.nlm.nih.gov/articles/PMC12133055/
- ZigZag QA. (2024). Revolutionizing Clinical Trials: ICH E6 R3 and Quality by Design. https://www.zigzagqa.com/resources/part-1-revolutionizing-clinical-trials-ich-e6-r3-and-quality-by-design/
- CCRPS. (2025). Medical Monitor Responsibilities in Clinical Trials. https://ccrps.org/clinical-research-blog/medical-monitor-responsibilities-in-a-clinical-trial
- Biomapas. (2025). Safety Monitoring In Early Phase Oncology Trials. https://www.biomapas.com/safety-monitoring-in-early-phase-oncology-trials/
- Southern Star Research. (2025). The Critical Impact of Medical Monitoring in Clinical Trials. https://southernstarresearch.com/news/the-critical-impact-of-medical-monitoring-in-clinical-trials/
- Schmidt, L., et al. (2024). Interactive medical and safety monitoring in clinical trials with reproducible data analysis workflows. Clinical Trials, 21(4), 467-476. https://pmc.ncbi.nlm.nih.gov/articles/PMC11271019/
- Clinical Leader. (2025). The New Era Of Medical Monitoring. https://www.clinicalleader.com/doc/the-new-era-of-medical-monitoring-0001
- Snapinn, S. M., et al. (2013). Safety Monitoring in Clinical Trials. Biopharmaceutical Report, 21(1), 2-14. https://pmc.ncbi.nlm.nih.gov/articles/PMC3834937/
- Intuition Labs. (2024). Good Clinical Practice (GCP): The 13 Principles Explained. https://intuitionlabs.ai/pdfs/good-clinical-practice-gcp-the-13-principles-explained.pdf
- European Medicines Agency. (2025). ICH E6 Good clinical practice - Scientific guideline. https://www.ema.europa.eu/en/ich-e6-good-clinical-practice-scientific-guideline
- Cyntegrity. (2026). ICH E6(R3) Sponsor Responsibilities: Active Quality Management. https://cyntegrity.com/ich-e6-r3-sponsor-responsibilities/
- Intuition Labs AI. (2024). Good Clinical Practice (GCP): The 13 Principles Explained - Quality Management Systems. https://intuitionlabs.ai/pdfs/good-clinical-practice-gcp-the-13-principles-explained.pdf
- Hunt, M. (2024). Part 1-Revolutionizing Clinical Trials: ICH E6 R3 and Quality by Design. ZigZag QA Resources. https://www.zigzagqa.com/resources/part-1-revolutionizing-clinical-trials-ich-e6-r3-and-quality-by-design/
- Morrison, B. W., et al. (2022). What is the purpose of clinical trial monitoring? A review of foundational principles. Clinical Trials, 19(5), 489-497. https://pmc.ncbi.nlm.nih.gov/articles/PMC9526458/
- Bersoff-Matcha, S. J., et al. (2001). Monitoring and ensuring safety during clinical research. JAMA, 285(9), 1201-1205. https://pubmed.ncbi.nlm.nih.gov/11231751/
- Novotech CRO. (2026). Professional Services for Your Early Phase Clinical Trials in Australia. https://novotech-cro.com/solutions/early-phase-trials-australia
- Novotech CRO. (2024). Medical Monitoring and Safety Services. https://novotech-cro.com
- EurekAlert. (2020). Novotech Research shows most Australian sites are open for clinical trials. https://www.eurekalert.org/news-releases/514441
- Datapharm Australia. (2023). Medical Monitoring and Pharmacovigilance Services. https://datapharmaustralia.com/services/medical-monitoring-and-pharmacovigilance-services
- Datapharm Australia. (2023). Clinical Trial Monitoring Services. https://datapharmaustralia.com/services/clinical-trial-monitoring-services
- Linear Clinical Research. (2025). Pioneer in Early-phase Clinical Trial Services in Australia. https://www.linear.org.au
- Sofpromed. (2022). Clinical Trial Monitoring Australia. https://www.sofpromed.com/monitoring-clinical-trials-australia
- Australian Healthcare Solutions. (2025). Clinical Trial Services | CRO. https://australianhealthcaresolutions.com.au/clinical-trial-services/
- Fishbein, G. (2022). De-risking Oncology Trials with Medical Monitoring. BioPharma Dive. https://www.biopharmadive.com/spons/de-risking-oncology-trials-with-medical-monitoring/626083/
- Pharmaceutical Technology. (2024). Data shows majority of Australian sites are open for clinical trial activity. https://www.pharmaceutical-technology.com/sponsored/
- Novotech CRO. (2024). Virtual Clinical Trials in Asia and Australia. https://novotech-cro.com/faq/virtual-clinical-trials-asia-and-australia
- European Medicines Agency. (2025). ICH E6 Good clinical practice - Overarching principles and Annex 1 adoption. https://www.ema.europa.eu/en/ich-e6-good-clinical-practice-scientific-guideline
About The Author:
 Dora V. Ardila, MBBS, Ph.D., MBA candidate, is a senior medical leader with over 10 years of international experience spanning clinical development, investigator-initiated and industry-sponsored trials, translational research, and healthtech advisory work. She has trained as a clinician-scientist at the Icahn School of Medicine at Mount Sinai and the University of Melbourne, with a Ph.D. completed in collaboration with the Victorian Comprehensive Cancer Center Alliance. Through her independent healthtech consulting practice, she partners with biotech, digital health, and AI-driven organizations to translate clinically rigorous innovation into scalable, ethically grounded solutions, advising on trial strategy, regulatory pathways, medical governance, and data-enabled decision-making. Her expertise encompasses protocol design, biomarker discovery, patient safety, regulatory compliance, cross-functional leadership, and enterprise-level medical strategy.
Dora V. Ardila, MBBS, Ph.D., MBA candidate, is a senior medical leader with over 10 years of international experience spanning clinical development, investigator-initiated and industry-sponsored trials, translational research, and healthtech advisory work. She has trained as a clinician-scientist at the Icahn School of Medicine at Mount Sinai and the University of Melbourne, with a Ph.D. completed in collaboration with the Victorian Comprehensive Cancer Center Alliance. Through her independent healthtech consulting practice, she partners with biotech, digital health, and AI-driven organizations to translate clinically rigorous innovation into scalable, ethically grounded solutions, advising on trial strategy, regulatory pathways, medical governance, and data-enabled decision-making. Her expertise encompasses protocol design, biomarker discovery, patient safety, regulatory compliance, cross-functional leadership, and enterprise-level medical strategy.
