Let's Tackle The Hidden Real-World Reasons For Poor Clinical Trial Diversity
By Eleanor Perfetto, Ph.D., and Jeanne M. Regnante

Disparities in clinical research participation are well documented and, like broader healthcare disparities, a byproduct of systemic and institutional racism. COVID-19 vaccine and treatment development have greatly amplified the need for all communities to have the same opportunity to participate in research. To achieve better outcomes and health equity for historically underserved populations, we must address the not widely acknowledged, hidden, real-world reasons for such poor diversity in clinical trials. The importance is clear: People’s lives depend on it. Here, we will pull back the covers on the issues we must tackle to improve diversity in trials.
Let’s tackle:
- Representativeness: The FDA routinely asks for a trial recruitment strategy consistent with the target disease population. Sponsors need to meet accrual milestones. Do you have a process for determining the numbers and types of “representative” populations for inclusion for U.S. trials?
- Clinical trial site placement: There is often a disconnect between clinical trial site placement and where patients with the disease reside, especially for racial and ethnic minority populations. Where should clinical trials be optimally placed so accrual of representative populations can be more easily achieved?
- Reimbursement costs for participants in clinical trials: What do study budgets pay for with consistency? Do you let patients know as part of expectations when recruiting? “It depends” is not a plan.
- Community and patient insights: Do you plan in advance and ask for input on the study question, eligibility criteria, and feasibility from diverse populations actually targeted to participate in the trial in the community where they live?
Tackling Representativeness1
“Representativeness” means a sufficient number of and types of people are included to ensure those enrolled represent the target population for whom the treatment is intended. It refers to “who” and “how many” individuals to include to, as closely as possible, represent the broader target patient population. To achieve representativeness:
- Define – Clearly define the trial and recruitment objective(s).
- Understand – Understand as much as possible about the full target population and any target subpopulation(s) and the challenges to reaching them.
- Specify – Develop a description of the minimum target(s) for representativeness for the trial.
- Plan – Develop a plan to achieve the minimum target(s).
- Evaluate – Develop an evaluation plan to assess progress on achieving target(s) or make adjustments if needed based on new information.
- Document – Record how patient representativeness was defined, targeted, achieved, and assessed.
“The drugs that we approve need to work for the majority of [the intended] patient population of use in the U.S.”
– Lola Fashoyin-Aje, MD, Ph.D. (FDA), ACCR panel, June 23, 2020
Recommendations: 2
- It is important for study sites, trial leaders, CROs, and operations personnel engaging with sites to know how the sponsor is defining goals for enrollment and representativeness for a given trial. (Note: Demographic representation objectives may not be in the protocol.)
- Optimal site planning depends on establishing and communicating objectives to all involved in trial recruitment. Don’t leave a discussion of your commitment to diversity to investigator communications only.
Tackling Clinical Trial Site Placement
We all realize there is urgency to improve clinical trial enrollment of racial and ethnic minority groups for equitable benefit of resulting innovation and access. Let’s start with better understanding the distribution and impact of a disease among racial and ethnic groups geographically.
Case in point: An analysis of the National Minority Quality Forum Lung Cancer Index3 (2017 Medicare FSS) data shows 77% of Medicare beneficiaries living with lung cancer reside in 20% of U.S. ZIP codes. Within this cohort, nine out of the top 10 states by total patient count also see highest patient counts for African American and Hispanic patients. With these data in hand, we asked: To what extent does the current U.S. clinical trial (CT) site placement (clinicaltrials.gov) for non-small cell lung cancer (NCSLC) and small cell lung cancer (SCLC) reflect the reality of lung cancer patient prevalence and demographics in the U.S.? Data suggest there is a major disconnect between U.S. lung CT placement and where diverse patient populations live.4
Tackling this issue requires a commitment to site planning, which may start with an overlap of prevalence of disease and demographics and current site placement.
Recommendations:
- Integrate data (prevalence of disease and demographics by ZIP code and county) from the National Minority Quality Forum Disease Indexes3 to inform the establishment of Diversity, Equity, and Inclusion Centers of Excellence among clinical trial investigative sites based on your therapeutic development plans.
- Inform the selection of new trial sites by considering publications and success factors/capablities to identify those sites with the ability to accrue diverse patients with high success.5,6
- This is a slow-motion tackle; a new site placement strategy takes planning, time, and long-term commitment.
Tackling Reimbursement For All Patients During Clinical Trials
The NMQF Diverse Cancer Communities Working Group survey (2019) asked about the degree to which major innovative cancer sponsors (N=6) have a standard approach to coverage or reimbursement of patients care during clinical trials. The results? “Well, it depends.”
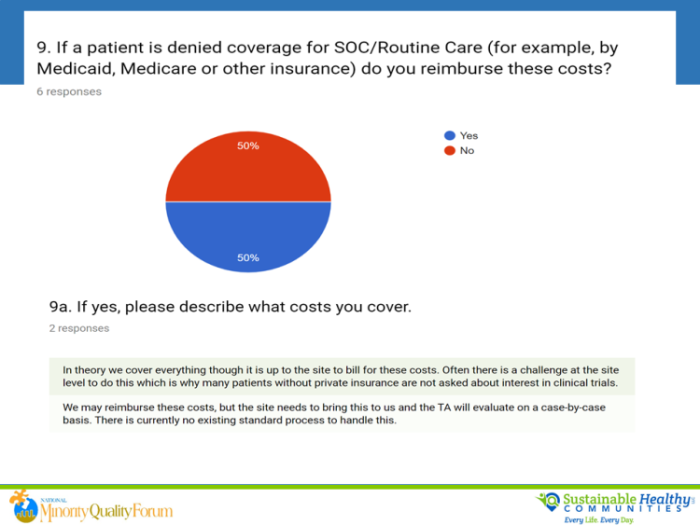
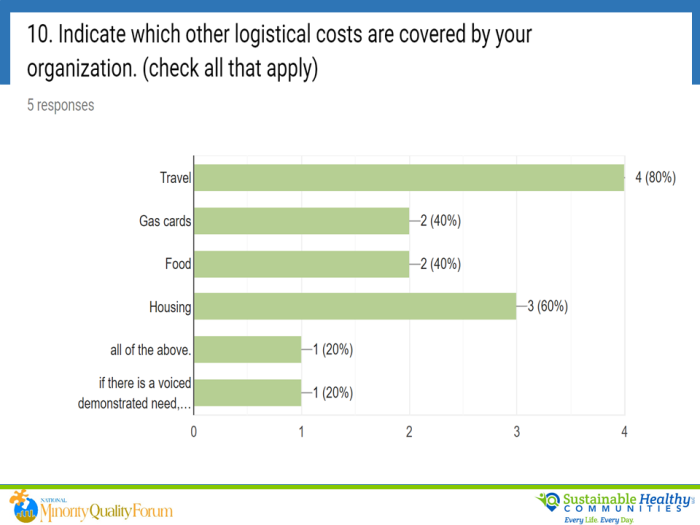
Appropriate reimbursement for patient logistical and medical expenses is a priority. It is well documented that populations with low socioeconomic status (SES) are not engaged in clinical trials in the U.S. Racial and ethnic minorities, and low SES Caucasians, are disproportionately affected. Limited engagement because of onerous compensation processes or policies is a trust issue. It amplifies barriers to achieve inclusion research goals. At the core, true engagement of diverse communities starts with fairness and equitable implementation. The FDA does not consider as undue influence reimbursement for reasonable travel expenses to and from the clinical trial site and associated costs such as airfare, parking, and lodging.7
We must start with appropriate coverage of costs that allows patients to participate. People should decide whether or not to be in a trial based on what it means to them, their family, and their community. Out-of-pocket expenses should not be a barrier to trial participation. Excessive or coercive compensation should, of course, be avoided. But we cannot achieve trial diversity and representativeness without appropriate, ethical reimbursement.8-12
Recommendations:
- Discuss with your IRB the importance of standardization of appropriate coverage of costs to patients.
- Cover all patient out-of-pocket expenses based on the principle that it should not cost the patient more to be in a clinical trial. This expectation should be discussed with the patient during the recruitment phase.
- Have explicit, written, approved policies with regard to reimbursement for trial participants; make the policies available to all trial sites and investigators to ensure study budgets and implementation practices are consistent with the guidance; include decision rights.
- Assume there are real costs to patients, families, and caregivers to engaging in clinical trials. Engage with diverse and vulnerable populations for input with regard to expenses and costs.
- Consider a prepaid card for all participants. This will help with efficient application of the policy, so it is not dependent on a patient asking for support or a clinical monitor making a determination in a timely manner.
Tackling Community And Patient Insights
Community engagement is a core function that drives trust, understanding, and access to care for racial and ethnic minority groups and their care partners. Early engagement of racial and ethnic minority and underserved populations at the community level is a proven enabler of access to care and participation in clinical trials throughout the cancer care continuum.
There is growing and impactful behavior by cancer centers to tailor interventions to the needs of vulnerable populations. The knowledge and trust gained by maintaining an ongoing relationship with community partners affords cancer centers the opportunity to tailor interventions to the needs of a vulnerable community and thereby increase the sustainability of efforts that influence patient behavior, leading to better patient outcomes. We reported previously that community engagement is a success factor for all cancer centers that are able to successfully accrue racial and ethnic minorities in clinical trials. In addition, in a recent research assessment focused on securing the cancer continuum of care model for racially and ethnically diverse and medically underserved populations, one of the highest cross-cancer-continuum areas of impact included excellence in community engagement as a necessary mandate to build trust among minority and underserved populations.13
In 2019, the lead author of a cancer center of excellence (COE) publications5,6 asked research leaders at COEs (N=4) what they need from sponsor partners to ensure racial and ethnic minorities are successfully included in clinical trials. (Note that at the time of this qualitative survey, there was an influx of requests from sponsors, midway through trials, asking for additional support from the centers for more inclusive populations for their ongoing studies.) In the section immediately below, we share the results of that survey — consensus advice from COE leaders at cancer centers in the U.S.
Recommendations:
- Make and communicate a clear assessment of the population of patients to be accrued in the trial based on the epidemiology of the disease; communicate recruitment and accrual goals and optimal timing with site leaders and with investigators.
- Tip 1: Provide justification of eligibility criteria and comorbidities for targeted demographics to be recruited based on expert input and data and/or ask sites for the optimal inclusion and exclusion criteria based on their experience.
- Tip 2: See the Tackling Representativeness section above.
- As part of deliberate planning for a trial, work with lead sites and ask their communities for input on the research question, protocol design, and study feasibility.
- Tip 1: This may be new for sponsors and should be planned well in advance with targeted study sites verses convening populations for broader insights when the broader groups may not be asked to participate in the trial at all.
- Tip 2: During protocol development, plan for mock trial enrollment at selected sites to gain patient and site reaction to and input on a protocol.
- Tip 3: Consider asking patient advocacy organizations to convene a community advisory board for patient insights with lead sites.
- Ask for input from the site operations and clinical investigators with respect to eligibility criteria reflective of the population trying to be reached.
- Tip 1: The sponsor should be open to aligning or amending the protocol to accommodate the population being sought.
- Tip 2: IRBs need to be educated as to what would be acceptable, but less restrictive, criteria.
- Discuss/deliver and support: health literate, culturally sensitive, and linguistically appropriate study recruitment materials for patients and their families. This includes such materials as: study summary, expectations for study participants and caregivers, and informed consent.
- Tip: The lead site should be identified to develop templates and customizable materials for all study sites.
- Discuss and create a budget and transparent process that accommodate logistic and medical support of patients. Discuss timely reimbursement of costs to patients (recommend prepaid cards) and allow for all eligible patients to be recruited regardless of insurance status.
- Tip: See Tackling Compensation For All Patients During Clinical Trials section above.
- Discuss and create a budget and timing for lay or clinical navigator(s) to focus on community outreach and outreach to PCP and other specialty networks (based on included comorbidities).
- Tip: It may be important to determine whether sites factor this capability into budget estimates for individual trials and how they prioritize allocation and timing of implementation of this capability.
Success tips are further documented with the publication of a multi-stakeholder effort with notable examples by the MRCT-Harvard.14
Implementation Success Tips For Sponsors
For each hidden real-world issue:
- Tackle it: Align on goals, timing, roles, decision rights with all stakeholders, including CROs, investigator sites, patient advocacy groups, etc., and ask them what they need from whom and when, to deliver on goals.
- Coordinate: Share the aligned process and roles with all stakeholders and operations. Ask that they put them into their plan and tell them why.
- Pilot: Pick a future trial for each real-world under-the-covers issue and pilot a process; ask all stakeholders how it went in your evaluation of the pilot.
- Share best practices: Report on successes via abstracts and multi-stakeholder communications; outline what it will take for your real-world practice to have national scalability, sustainability, and impact with strong dissemination.
- Continuous improvement depends on uncovering additional “hidden real-world issues” and continued learning and improvement.
Continued focus and progress on increased participation of racial and ethnic minorities in clinical research is needed from multiple collaborative stakeholders to continue to improve our understanding of differences in risk and disease outcomes across populations … so let’s keep tackling together.
References:
- Tackling Representativeness: A Roadmap and Rubric, National Health Council, 2019. https://nationalhealthcouncil.org/wp-content/uploads/2019/12/Representativeness%20in%20Patient%20Engagement.pdf ;
- Enhancing the Diversity of Clinical Trial Populations — Eligibility Criteria, Enrollment Practices, and Trial Designs, FDA Guidance for Industry https://www.fda.gov/regulatory-information/search-fda-guidance-documents/enhancing-diversity-clinical-trial-populations-eligibility-criteria-enrollment-practices-and-trial
- National Minority Quality Forum (NMQF) Lung Cancer Index 2017 Medicare FSS [Database] https://mypalc.org/
- Roy UB, et al - Geographic relationship between lung cancer clinical trial sites and patient prevalence and demographics in the Medicare Fee-for-service program, AACR The Science of Cancer Health Disparities in Racial/Ethnic Minorities and the Medically Underserved, Proceedings, Sept 20-23, 2019, San Francisco, CA
- Regnante JM, Richie NA, Fashoyin-Aje L, Vichnin M, Ford ME, Roy UB, Turner K, Hall LL, González ET, Esnaola NF, Clark LT, Adams HC III, Alese OB, Gogineni K, McNeil LH, Petereit DG, Sargeant I, Dang JH, Obasaju C, Highsmith Q, Craddock Lee SJ, Hoover SC, Williams EL, Chen MS Jr.: US Cancer Centers of Excellence strategies for increased inclusion of racial and ethnic minorities in clinical trials. J Oncol Prac DOI 10.1200/JOP.18.00638 http://ascopubs.org/doi/full/10.1200/JOP.18.00638 (EDITORS PICK AWARD 2019)
- Regnante J, Richie N, Fashoyin-Aje L, Hall LL, Highsmith Q, Louis J, Turner K, Hoover S, Lee S, González E, Williams E, Adams H III, Obasaju C, Sargeant I, Spinner J, Reddick C, Gandee M, Geday M, Dang JT, Watson R, Chen M Jr., Operational strategies in US cancer centers of excellence that support the successful accrual of racial and ethnic minorities in clinical trials, Contemporary Clinical Trials Communications https://doi.org/10.1016/j.conctc.2020.100532
- FDA Guidance on out- of- pocket costs https://www.fda.gov/regulatory-information/search-fda-guidance-documents/payment-and-reimbursement-research-subjects
- https://www.eupati.eu/clinical-development-and-trials/compensation-clinical-trials/#References
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3601710/
- https://www.pfizer.com/research/research_clinical_trials/compensation_trial_participants
- https://forteresearch.com/news/fda-specifies-patient-reimbursements-vs-compensation-encourage-recruitment-equity-clinical-trials/
- https://www.centerwatch.com/articles/12708-fda-clarifies-stance-on-clinical-trial-reimbursements-for-patient-travel-lodging
- Winkfield K, Regnante JM, Sonet E, González E, Freund K, Doykos P, on behalf of the Cancer Continuum of care for Medically Underserved Populations Working Group. Development of an actionable framework to address cancer care disparities in medically underserved populations in the United States: expert roundtable recommendations, IN PRESS, November 2020, Journal of Oncology Practice
- Bierer B.E., White S.A., Meloney L.G., Ahmed H.R., Strauss D.H., Clark L.T., (2020) Achieving Diversity, Inclusion, and Equity in Clinical Research Version 1.0. Cambridge and Boston, MA: Multi-Regional Clinical Trials Center of Brigham and Women’s Hospital and Harvard (MRCT Center). Retrieved from: https://mrctcenter.org/diversity-in-clinical-trials/
About the Authors:
 Eleanor Perfetto, Ph.D., is the interim CEO of the National Health Council (NHC). She was named SVP of strategic initiatives for the NHC in July 2015 and was promoted to EVP in January 2019. She also holds a part-time faculty appointment at the University of Maryland, Baltimore School of Pharmacy, where she is professor of Pharmaceutical Health Service Research. Her research and policy work primarily focus on patient engagement in comparative effectiveness and patient-centered outcomes research; medical product development; patient-reported outcome selection and development; and healthcare quality. Perfetto holds BS and MS degrees in pharmacy from the University of Rhode Island, and a Ph.D. from the University of North Carolina School of Public Health with concentrations in health policy and epidemiology.
Eleanor Perfetto, Ph.D., is the interim CEO of the National Health Council (NHC). She was named SVP of strategic initiatives for the NHC in July 2015 and was promoted to EVP in January 2019. She also holds a part-time faculty appointment at the University of Maryland, Baltimore School of Pharmacy, where she is professor of Pharmaceutical Health Service Research. Her research and policy work primarily focus on patient engagement in comparative effectiveness and patient-centered outcomes research; medical product development; patient-reported outcome selection and development; and healthcare quality. Perfetto holds BS and MS degrees in pharmacy from the University of Rhode Island, and a Ph.D. from the University of North Carolina School of Public Health with concentrations in health policy and epidemiology.
 Jeanne M. Regnante is the chief health equity and diversity officer at the LUNGevity Foundation. In this role, her primary objective is to secure the engagement and insights of vulnerable lung cancer communities by working with trusted community-based organizations and multiple healthcare stakeholders to achieve health equity and equalize disparities for all. Previous to her current position, Regnante was SVP of community engagement for the National Minority Quality Forum (NMQF), where she chaired the ongoing and impactful Diverse Cancer Communities Working Group (CWG). She also spent 30+ years at Merck & Co., where she served as head of global patient engagement, chief of staff to Merck’s chief medical officer, and head of scientific affairs operations and led a company-wide strategy focused on diversity and inclusion in clinical research.
Jeanne M. Regnante is the chief health equity and diversity officer at the LUNGevity Foundation. In this role, her primary objective is to secure the engagement and insights of vulnerable lung cancer communities by working with trusted community-based organizations and multiple healthcare stakeholders to achieve health equity and equalize disparities for all. Previous to her current position, Regnante was SVP of community engagement for the National Minority Quality Forum (NMQF), where she chaired the ongoing and impactful Diverse Cancer Communities Working Group (CWG). She also spent 30+ years at Merck & Co., where she served as head of global patient engagement, chief of staff to Merck’s chief medical officer, and head of scientific affairs operations and led a company-wide strategy focused on diversity and inclusion in clinical research.
