Redefining Patient Outcomes Measurement Using RWE Data
By Monica Nandagopal, senior analyst, Beroe Inc.

Patient outcomes measurement is undergoing a transformation as healthcare systems shift from controlled environments toward real-world, value-based decision-making. While RCTs remain the standard for establishing safety and efficacy, their single time measurement-based endpoints often fail to capture how therapies perform across diverse populations, routine clinical settings, and longitudinal patient journeys. RWE redefines patient outcomes measurement into a continuous, patient-centered, and decision-grade framework that reflects patient journey, quality of life, treatment durability, and economic impact over time. This article proposes an outcome-channeling framework that organizes patient outcomes into multidimensional domains and emphasizes longitudinal trajectories rather than single time endpoints.1,2
Limitations Of Traditional Patient Outcomes Measurement
Traditional outcome measurement approaches are often:
- Generic datapoints: Clinical trials frequently rely on laboratory datapoints, radiographic changes, or intermediate endpoints that do not fully reflect daily functioning, symptom burden, or quality of life.3
- Episodic and time-point based: Outcomes are measured at predefined milestones (e.g., six or 12 months), failing to capture dynamic treatment trajectories, disease fluctuations, and adherence patterns.3
- Limited inclusion of social determinants: Traditional metrics ignore contextual factors, such as socioeconomic status, access to care, geographic barriers, and health literacy.4,5
- Weak in reflecting daily function and health states: Event-based measures (e.g., hospitalization, relapse) overlook functional parameters, symptom variability, and patient-reported experiences, thereby underrepresenting the disease burden.4
These limitations create a gap that constrains clinical decision-making.
A Better Framework For Measuring Patient Outcomes Using RWE
Traditional clinical trials measure outcomes using single static endpoints. However, real patients experience disease and treatments dynamically.5
Therefore, the proposed framework is a patient-centered, continuous outcome measurement model using RWE derived from multiple healthcare data sources. The model emphasizes multidimensional outcomes, longitudinal patient trajectories, integrated data ecosystems, and composite outcome indices to support decision-grade healthcare insights.4,5
Multidimensional Outcome Framework
Real patient outcomes involve multiple dimensions, such as survival, quality of life, treatment burden, and healthcare utilization.5,6
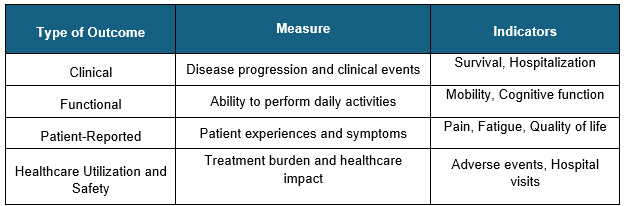
Sources: Beroe Analysis, Secondary Sources
Patient-reported outcomes are increasingly integrated into care to capture symptoms and quality-of-life impacts that are not visible in clinical records.6
Emphasis On Outcome Trajectories
Because chronic diseases evolve continuously and treatment responses change over time, the below outcome trajectory tracks how a patient's health status evolves.
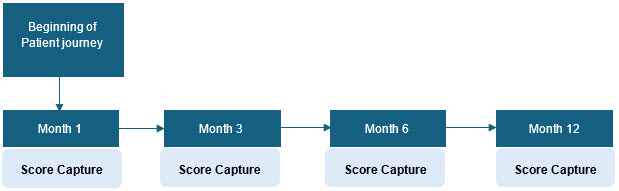
Sources: Beroe Analysis, Journals
Longitudinal EHR data allows researchers to analyze disease progression and treatment outcomes across time.5,6
Heterogeneous Data Integration And Weighting Schemes
Evaluating multiple RWD sources allows researchers to obtain an outcome metric score of patient journey, linking treatment patterns with clinical outcomes and healthcare utilization.6,7
Multiple data sources that could be integrated include:
- EHRs
- claims data
- laboratory systems
- pharmacy data
- registries
- patient-reported data.8
For example: Outcome Score = (Weightage for Clinical × Clinical outcomes) + (Weightage for functional × Functional outcomes) + (Weightage for patient outcomes × Patient-reported outcomes) + (Weightage for safety × Safety/utilization)8
These weights ensure the index reflects true clinical importance and patient priorities.
Building An End-to-End Framework Architecture
It is essential to build an end-to-end framework for using these RWD sources and integrating them in the proposed framework. This outcome framework leads to dynamic patient trajectories by integrating multiple RWD sources and constructing multi-domain composite outcome measures.8,9
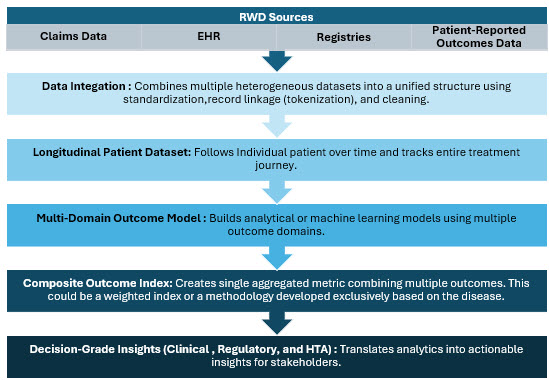
Sources: Beroe Analysis, Quality of Life Research
This approach enables:
- earlier identification of treatment response,
- more patient-centered outcome measurements, and
- better support for regulatory, clinical, and health technology decision-making.9,10
Methodical Shifts Required To Enable RWE-Driven Outcomes
Regulators and HTA bodies emphasize that decision-grade RWE depends on both data reliability/relevance and fit-for-purpose methods.8,9 Effective methods that could bring strong RWE driven outcomes include:
- Use of Outcome Trajectories: Capturing continuous records of outcome trajectories and evolution would lead to understanding a patient’s journey better.8 Collecting outcome signals with time-stamped measurements from data sources and constructing trajectory metrics using interpretable measures, such as slope of improvement or decline or area under the curve (AUC), serve as essential methods of implementation.9
- Adapting to RWD realities: RWD often contains incomplete records, inconsistent coding practices, and irregular follow-up intervals. To produce scientifically credible evidence, researchers must ensure data quality and consistency before analysis.11 Standardized coding systems will harmonize procedure and medication codes. Managing missing data using predefined approaches such as multiple imputation, sensitivity analyses, and robust statistical models leads to better data accuracy.
- Synthesis of multi–source outcome construction: A multi-source synthesis approach integrates different data streams into a single longitudinal patient record, enabling a more comprehensive view of disease progression and treatment outcomes.8 Some techniques of implementation include establishing patient-level linkage using deterministic or probabilistic matching approaches, defining the source that is essential for diagnosis assessment, hospitalization, mortality, or symptom burden, and constructing composite outcomes where necessary, using multiple contributing datapoints rather than one field.11
- Patient-Centered Outcome Domains: RWE-driven outcomes models expand to include patient-centered domains, such as symptom burden, daily functioning, quality of life, treatment burden, caregiver dependence, and ability to participate in normal activities.4,5 Select validated instruments and define real-world points where direct measures do not exist and create integration between clinical events and patient-centered states that improve interpretability.4
- Technology-Driven Outcome Measurement: Technology enables scalable evidence–driven outcome measurement by integrating diverse healthcare data sets and innovative models.12,13 One implementation technique includes using natural language processing to extract clinically relevant signals, such as symptoms, disease severity, and adverse events, from physician notes and narrative reports, wearable devices, and mobile health platforms to capture continuous patient activity.12 Causal and comparative effectiveness analytics apply advanced statistical approaches, such as propensity score matching (comparing outcomes between patients who are matched using probability), inverse probability weighting (different importance [weights] to patients so that the treatment groups become balanced), and target trial emulation (designing an observational study to mimic the structure of a randomized clinical trial).13
Case Example Illustrating Patient Outcomes Using RWE
The following pharma companies illustrate the use of technology and innovative methodologies to build effective trial results.
- Ametris DECODE Program: Advancing Wearable-Based Patient Outcomes in Obesity Trials
- Ametris, through its DECODE Obesity, collaborates with Novo Nordisk, Eli Lilly, Roche, and AstraZeneca and is developing digital endpoints based on continuous wearable measurements of physical activity, functional mobility, and sleep, moving beyond traditional clinic-based measures such as weight or periodic functional tests.
- The methodology combines continuous wearable monitoring, integrated eCOA platforms (via its partnership with Kayentis) that link patient questionnaires with device data, and adherence management tools such as real-time reminders and regulatory-aligned evidence generation frameworks to produce validated digital outcome measures.14,15,16
- BD Libertas wearable injector that enables patient-centered therapy delivery outcomes
- BD launched the first pharma-sponsored clinical trial using the BD Libertas wearable injector for subcutaneous delivery of complex biologic drugs.
- The wearable injector is a prefilled, ready-to-use system designed to deliver high-viscosity biologics via subcutaneous injection, enabling therapies that normally require hospital infusions to be administered through self-injection at home.
- The trial builds on more than 50 preclinical and clinical studies, including a device study where 100% of participants indicated they would likely use the injector if prescribed, highlighting strong patient acceptability.17
By integrating diverse real-world data sources, applying robust analytical methods, and leveraging digital health technologies such as wearables and mobile platforms, healthcare stakeholders can capture outcomes that better reflect real patient experiences and treatment effectiveness. Implementing transparent methodologies, strengthening data infrastructure, and encouraging cross-industry collaboration will be essential to generate credible, decision-grade evidence that supports regulatory, payer, and clinical decision-making while ultimately improving patient care.4,5,8,9
References:
- “Real-World Data: Assessing Electronic Health Records and Medical Claims Data to Support Regulatory Decision-Making for Drug and Biological Products,” July 2024. [Online]. Available: https://www.fda.gov/media/152503/download.
- “NICE real-world evidence framework,” June 2022. [Online]. Available: https://www.nice.org.uk/corporate/ecd9/resources/nice-realworld-evidence-framework-pdf-1124020816837.
- “Clinical Outcome Assessments (COAs) in Medical Device Decision Making,” [Online]. Available: https://www.fda.gov/about-fda/division-patient-centered-development/clinical-outcome-assessments-coas-medical-device-decision-making.
- “FDA Patient-Focused Drug Development Guidance Series for Enhancing the Incorporation of the Patient’s Voice in Medical Product Development and Regulatory Decision Making,” [Online]. Available: https://www.fda.gov/drugs/development-approval-process-drugs/fda-patient-focused-drug-development-guidance-series-enhancing-incorporation-patients-voice-medical.
- E. J. Pérez-Stable and S. N. Zenk, “Advancing Social Determinants of Health Research at NIH Through Cross-Cutting Collaboration,” 17 July 2023. [Online]. Available: https://www.ninr.nih.gov/newsandevents/news/advancing-social-determinants-health-research-nih-through-cross-cutting.
- “Core Patient-Reported Outcomes in Cancer Clinical Trials,” October 2024. [Online]. Available: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/core-patient-reported-outcomes-cancer-clinical-trials.
- F. Huang, J. Hou, N. Zhou, K. Greco, C. Lin, S. M. Sweet, J. Wen, L. Shen, N. Gonzalez, S. Zhang, K. P. Liao, T. Cai, Z. Xia, F. T. Bourgeois and T. Cai, “Advancing the Use of Longitudinal Electronic Health Records: Tutorial for Uncovering Real-World Evidence in Chronic Disease Outcomes,” J Med Internet Res, p. 27, 12 May 2025.
- G. A. Wells, F. Guillemin, P. A. Merkel, M. Wit, S. Mackie, L. March, G. Tómasson, L. K. King, S. M. Cembalo, S. Grosskleg, L. J. Maxwell, S. Monti, K. A. Quinn, B. J. Shea, P. Tugwell and D. Beaton, “Advancing composite outcome measures: Insights on weighting components from OMERACT 2023,” Seminars in Arthritis and Rheumatism, vol. 69, p. December, 2024.
- L. Schöner, D. Kuklinski, A. Geissler, R. Busse and C. Pross, “A composite measure for patient reported outcomes in orthopedic care: design principles and validity checks,” Quality of Life Research, 2023.
- M. Vargas-Fernández, J. d. D. M.-M. J. Luna and P. Carmona-Sáez, “Network analysis of longitudinal electronic health records using linear mixed models,” BioData Mining, 2026.
- “NICE real-world evidence framework: Assessing Data Suitability,” June 2022. [Online]. Available: https://www.nice.org.uk/corporate/ecd9/chapter/assessing-data-suitability.
- “Considerations for the Use of Artificial Intelligence To Support Regulatory Decision-Making for Drug and Biological Products,” January 2025. [Online]. Available: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/considerations-use-artificial-intelligence-support-regulatory-decision-making-drug-and-biological.
- P. S. Shirley V. Wang and M. S. Sebastian Schneeweiss, “Emulation of Randomized Clinical Trials With Nonrandomized Database Analyses,” Journal of the American Medical Association, 2023.
- “Kayentis and Ametris join forces to deliver integrated eCOA solution measuring and improving wearable technology adherence in clinical trials,” 24 February 2026. [Online]. Available: https://kayentis.com/resources/kayentis-and-ametris-join-forces-to-deliver-integrated-ecoa-solution-measuring-and-improving-wearable-technology-adherence-in-clinical-trials/.
- “Ametris starts obesity digital endpoint project with pharma,” September 2025. [Online]. Available: https://pharmaphorum.com/news/ametris-starts-obesity-digital-endpoint-project-pharma.
- “Ametris Expands Collaborative Research Project to Investigate Digital Measures of Physical Function for Drug Development in Obesity,” 02 September 2025. [Online]. Available: https://blog.ametris.com/news/ametris-decode-obesity.
- “BD Announces First Pharma-Sponsored Clinical Trial Using BD Libertas™ Wearable Injector Technology for Biologic Drugs,” 23 July 2025. [Online]. Available: https://investors.bd.com/news-events/press-releases/detail/894/bd-announces-first-pharma-sponsored-clinical-trial-using-bd-libertas-wearable-injector-technology-for-biologic-drugs.
About The Author:
 Monica Nandagopal is a senior research analyst with over eight years of experience in market research and consulting. Her insights have enabled top pharma companies in their strategic decisions on supplier outsourcing, category management, and planning. In the past year, she was engaged in multiple market sourcing studies, supplier data visualizations, and multiple quick reactive analysis across clientele for global and regional requirements.
Monica Nandagopal is a senior research analyst with over eight years of experience in market research and consulting. Her insights have enabled top pharma companies in their strategic decisions on supplier outsourcing, category management, and planning. In the past year, she was engaged in multiple market sourcing studies, supplier data visualizations, and multiple quick reactive analysis across clientele for global and regional requirements.
