Small Biotech Survival: Navigating Risk And Capital Scarcity With AI
By Maxine Opperman

The clinical development pathway for regulatory approval and beyond for small biotech companies has become ever more challenging. Rising clinical trial costs, enrollment difficulties, vendor failures, and the increase in site technical challenges necessitate new tools to overcome the rising risks in this changing environment. What has become obvious is that companies that do not employ forms of AI into their work processes will eventually lose productivity relative to those who do. This will be especially true for the small biotech that depends on a talented but small core team with extensive outsourcing.
Small Biotech Pros, Cons, And Risk Factors
Big Pharma has deep pockets. Even the midsize companies can weather failures, but the small biotech that operates on slim margins or in the red for decades relies on investor confidence and goodwill. One bad outcome can deliver existential failure.
Focusing on the smaller biotech company with market capitalization or private valuation typically through $5 billion, the challenges are immense. Market cap is a fluid measure of investor confidence or perception, not necessarily a rigorous picture of company scale, revenue, or potential. A company’s fundamentals or technicals are important, but for biotechnology companies, especially the smaller ones, it can be misleading and insufficient compared with other sectors, even pharma companies. Less tangible assets, such as intellectual property (IP), are not a component of market cap. Most, if not all, of these companies have no earnings and limited revenues rendering such measurements questionable.
The small biotech leverages several key advantages and a myriad of challenges, outlined, generally, below. The explosion in costs to run clinical trials today is well documented and becoming ever more burdensome for the small biotech operating with limited resources.
Table 1: Characteristics of the Small Biotech
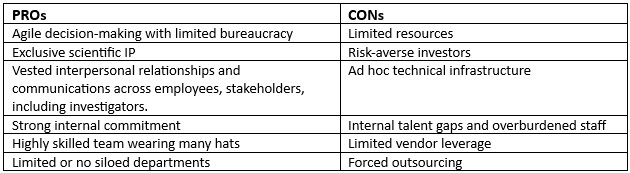
Emerging biotech companies engaged in R&D exclusively and those in early commercialization are both enmeshed in high risk but for different reasons. High risk often translates into incurring a high cost for needed capital where funding is typically tied to successfully meeting specified milestones. A continued inflow of capital from investors is needed to move forward. This creates challenges in securing ongoing funding when there are unanticipated delays and expenses. Suffering runway depletion beyond scope can become an ever more challenging reality.
Exceeding cash runway, the available number of months to operate without new capital infusion, can occur for a variety of reasons with serious implications on investor certainty, the cost of capital, and overall risk. When engaged in R&D exclusively, study-related factors impacting risk directly include:
- efficacy failure
- toxicity safety signals
- clinical trial delays caused by
- site competition
- failure to achieve enrollment within expected timelines
- excessive screen failure rates (SFR)
- patient retention issues
- high amendment rates
- vendor failures.
According to the Tufts Center for the Study of Drug Development, in 2021 the average direct cost of running Phase 2 or 3 studies was $40,000 per day. Further, the cost of each screen failure across all phases has been estimated to range from $5,000 to $12,000.
Another measure of risk, the likelihood of approval (LOA), revealed only a 60 percent likelihood as measured across all indications for Phase 1 through Phase 3 studies in 2021. Phase 2 to 3 studies scored the lowest LOA with 28.9 percent.¹
Early commercialization companies suffer different risks and challenges having successfully emerged with one regulatory approval. Exceedingly large amounts of capital are required to launch. Surviving the “fourth hurdle,” or securing healthcare payer reimbursement, means not only achieving efficacy and safety in the manufacturing process (GMP) but also delivering quality and value. includes assigning a price that effectively balances company costs against what payers are willing to pay, and payer reimbursement is not guaranteed. It will depend upon whether similar or existing treatments are available that are less costly or preferred.² Once a drug gains regulatory approval, the fourth hurdle is a vital step in commercialization, obtaining healthcare payer reimbursement. Additional challenges can occur that threaten survivability:
- failure to meet targeted sales revenue streams
- inability to successfully meet commercialized manufacturing demands
- failure to achieve bioequivalence (BE)
The observed process of moving from dependence on investor capital to approaching profitability is anything but certain. For example, consider the epic failure to commercialize plazomicin by Achaogen. The antibiotic plazomicin was approved in 2018 for the treatment of complex UTIs. Achaogen was a small biotech that developed the treatment, but was unable to fund commercialization due to perceived high-risk factors. Forced into bankruptcy in 2019, it was sold in auction to Cipla USA where the drug was later marketed under the name Zemdri.
This does not mean that risk becomes insurmountable for all recent approvals. In fact, the cost of capital should decline post-approval, but the amount required to launch exceeds previous study costs.
The underlying concern of getting treatment to patients in need as quickly as possible cannot be minimized. The time gap between regulatory approval and delivery to patients can mean lives lost. The longer it takes, the more cash burn. Unplanned/unexpected delays mean heightened uncertainty to investors.
Any or all of these factors impact a company’s risk profile with varying degrees of severity increasing the cost and access to capital. However, once regulatory approval is achieved, many risk factors decline.
Escalating Costs Of Running Trials
Amidst existing challenges of securing ongoing needed funding, the cost of running studies continues to increase. Deloitte reported the average cost per pipeline asset for R&D in the pharmaceutical industry was $2.23 billion in 2024 compared with $1.3 billion in 2013, showing a 72 percent increase. This measure of cost covers drug discovery through to launch. ⁴
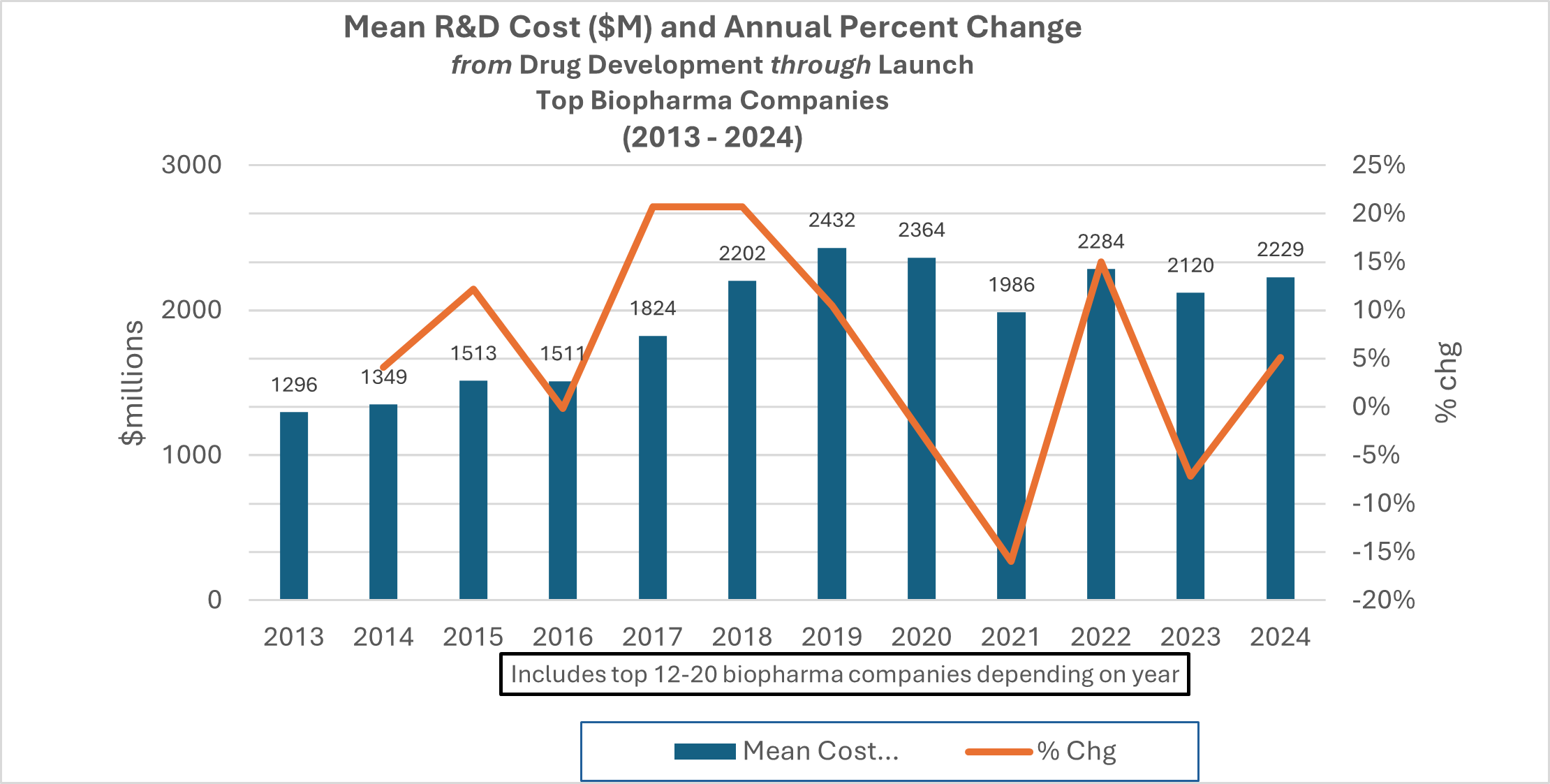
Source: Chart derived from Deloitte, March 2025 ⁴
Primary factors and obstacles contributing to increased costs are consistent with trial runway depletion reasons already mentioned. More recent factors adding to cost include changes in regulatory requirements, increased complexity of study designs, and the additional duration required, particularly among Phase 3 studies, to accommodate these complexities. Related to this is the increased number of data points collected, particularly in oncology studies, and extended data cleaning timelines potentially delaying data lock and analysis. Orphan drug therapies also have significantly smaller patient populations, placing additional challenges on finding patients and meeting enrollment requirements.
For the small biotech, avoiding the fourth hurdle altogether, instead of launching into commercialization with its immeasurable uncertainties, has been achieved through licensing, partnering, collaborating, or selling majority shares, and it has increased enormously.
Big Pharma has shifted its approach in recent years. Approvals among the top 20 biopharma companies for new molecular entities (NMEs) and new therapeutic biologics (NTBs) found 41 percent associated with external mergers and acquisitions and 25 percent via external licensing agreements from 2015 – 2021.⁵ The effort by larger companies to acquire and consolidate is only expected to increase in 2026-2030. This is especially true in specific therapeutic areas, modalities, and specialized platforms and technologies. Small biotech companies are being bought up in record number.
Big Pharma acquiring innovative small biotechs in late-stage development and early commercialization increases their pipeline and potentially averts some of the impending revenue losses anticipated from upcoming patent expirations and LOE. The patent cliff represents a massive exposure for Big Pharma to the tune of $200 billion to $400 billion. Their blockbuster drugs will no longer generate the millions of dollars in revenue as biosimilars and generics become available from 2026-2030. Later-stage acquisitions by Big Pharma offer lower risks comparatively. However, early-stage biotechs with drug discovery platforms are also being acquired in record number to revitalize innovation at large pharmaceutical companies.
How Can AI Help Small Biotechs?
How wisely and strategically AI is integrated into business processes will clearly impact ROI. Ensuring human oversight is vital for success but certainly not sufficient.
Even with meticulous evaluation and planning, serious setbacks can ultimately result, which raises two key questions:
- Why do most AI piloted projects fail?1
- Can the small biotech continue to exist in the market without employing AI?
Cost/Benefit
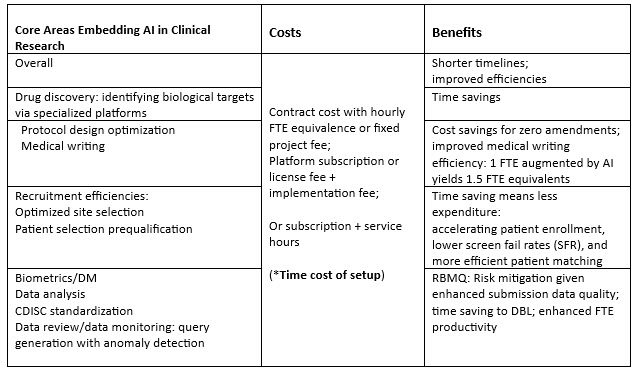
The costs and benefits of AI implementation are evaluated across two options. The benefits are reduced timelines that translate into decreased costs. One option is an outsourced model using functional service providers (FSPs) with AI-targeted services. Specialized FSPs offer niche AI-services across one or several core workflows.
Some FSPs offer flexible models depending on the needs, objectives, and existing internal competencies of the sponsor and can include:
- fully managed by the FSP
- sponsor operated independently
- hybrid collaboration.
Financial costs may include a premium associated with utilizing the FSP’s AI-services. The benefits are time-saving efficiencies with improved quality results and enhanced productivity for internal talent. Repetitive tasks are transferred to AI agents.
Many of the services and core workflows currently available are well documented.
Table 2: Outsourcing to an FSP with AI-targeted services
FSP Management
Employing a proven FSP and contractually building in predefined milestones can transfer the perceived risk associated with utilizing AI to the vendor for the specific study, but it won’t give you the ability to transform your organization into an AI-capable one. There are still risks in utilizing an FSP, and the setup time may obscure ROI. The study itself may be completed before long-term ROI is realized.
AI implementation from a regulatory perspective provides reduced labor costs and delivers faster approvals. More specifically, reducing manual medical writing, improving compliance through quality oversight, and reviewing and implementing regulatory changes as they occur all reduce timelines and lower costs.
In-House AI Investment
A second option is for the sponsor to invest in AI directly by building AI processes internally to replace manually-completed repetitive tasks and incorporating them into workflows. Contracting with a cloud-based data platform would be required and would facilitate needed data transformations. Small incremental investment building on successes can be a starting point. This is not without costs and requires more than a little groundwork. It also requires company-wide commitment.
According to most experts, building the necessary architecture from the ground up with the ongoing ability to scale are basic requirements for a long-term successful AI implementation and utilization.
- Building internal expertise
- Evaluating all internal processes and workflows
- Identifying existing hardware deficiencies
- Clearly defining goals
- Rebuilding all workflows
- Restructuring data and data systems
- Building governance and documentation
AI projects often fail because all factors are not considered and/or the initial architecture required is compromised. Scaling, the ability to implement across variations, must be implicit in the design.
Will you rely on external experts exclusively, or will you use and exploit the knowledge offered by FSPs while, at the very least, scoping your current systems and building internal expertise in parallel?
The small biotech sector drives drug discovery and innovation, and there is no longer any margin of error in their operations. By embedding AI into the clinical lifecycle, these companies can survive amid spiraling costs and burn rates of $40,000 per day.
But in order to transform years into months or even weeks in drug discovery, eliminate multiple amendments, reduce recruitment delays, and lessen vendor failures, they’ll still need to operationalize processes that eliminate manual data management. AI has become the key that will allow the small biotech to reach the next milestone, secure the next round of funding, and deliver life-saving therapies to market.
References
- BIO | QLS Advisors | Informa UK Ltd 2021, Clinical Development Success Rates and Contributing Factors 2011-2020 Report, https://www.bio.org/clinical-development-success-rates-and-contributing-factors-2011-2020
- Rawlins, Michael D. Crossing the fourth hurdle, Br J Clin Pharmacol 73:6, (07Mar2012)
- Wells, N., Nguyen, VK. & Harbarth, S. Novel insights from financial analysis of the failure to commercialise plazomicin: Implications for the antibiotic investment ecosystem. Humanit Soc Sci Commun 11, 941 (2024). https://doi.org/10.1057/s41599-024-03452-0.
- Deloitte, Measuring the Return from Pharmaceutical Innovation 15th Edition (March 2025)
- Schuhmacher A, Hinder M, Dodel A, Gassmann O, Hartl D. Investigating the origins of recent pharmaceutical innovation. Nat Rev Drug Discov. 2023 Oct;22(10):781-782. doi: 10.1038/d41573-023-00102-z. PMID: 37407752.Nature Reviews, Supplementary information table 4
- MIT Media Lab, The GenAI Divide: State of AI in Business (July 2025). https://cloudelligent.com/wp-content/uploads/2026/02/v0.1_Stateof_AI_in_Business_2025_Report.pdf.
About The Author:
 Maxine Opperman has been working in clinical data management for over 25 years. She has experience working in clinical research in non-profit, for multiple CROs, and on the sponsor side. For the past 9 years, she has been working for Mesoblast, , in the development of cellular medicines for the treatment of inflammatory conditions. She has a BA and MA (ABT) in economics.
Maxine Opperman has been working in clinical data management for over 25 years. She has experience working in clinical research in non-profit, for multiple CROs, and on the sponsor side. For the past 9 years, she has been working for Mesoblast, , in the development of cellular medicines for the treatment of inflammatory conditions. She has a BA and MA (ABT) in economics.
