
DATA MANAGEMENT
Harnessing The Potential Of Patient-Level Data In Clinical Trials
Join our panel of industry leaders as they explore innovative methods and best practices for incorporating fully consented patient data in clinical trials.
Leveraging DHTs To Improve Data Collection And Analysis
How can wearable DHTs be leveraged to measure physical activity in patients with immunological diseases, enhancing clinical trials and advancing drug development?

Quality Insights For Ophthalmology Innovation
Verana Health provides data essential to answering your questions during the drug development process, particularly in the field of ophthalmology.

Trialtrove: Clinical Trial Data You Can Depend On
Trialtrove’s data, compiled from over 58,000 sources, helps study sponsors and CROs make informed decisions around clinical trial strategy, design, and execution.

Medidata Clinical Data Management
By automating complex tasks, Medidata Rave simplifies clinical data management and capture. This leads to cleaner, more reliable data, allowing for quicker analysis and results.

Addressing Opportunities And Challenges In Digital Health Data
Experts from diverse industry sectors unpack key points from the FDA’s final guidance on Digital Health Technologies (DHT) for remote data collection in clinical investigations.

The Impact Of AI On Mobile Visits
Experts discuss the FDA's new AI guidance, global harmonization, and the ethical implications of AI in clinical trials. Discover the challenges and opportunities ahead.

Combining De-Identified EHR And Claims Data
Learn how EHR+ Data can improve your understanding of the patient's journey in ophthalmology, urology, and neurology clinical trials.

Yes, Your Study Is At Risk: Risk Mitigation Plans For Endpoints
Ocular endpoints often introduce unique bottlenecks that can stall enrollment and compromise data integrity. This presentation provides the tools for a streamlined, resilient clinical trial strategy.

Keeping Up With ClinOps: Why And How To Improve Delivery
Industry experts discuss the concept of a trial platform as a service and how it can accelerate and enhance the day-to-day functions of clinical operations throughout the entire lifecycle of a study.

Medidata Research Alliance
The Medidata Research Alliance collaborates with scientific and medical research communities to use clinical trial data for medical advancements and increased patient access to innovative treatments.

Manage Exponential Data Growth With Medidata Clinical Data Studio
Bring study teams together like never before for total data quality oversight.

Global Compliance Made Easy
Global clinical trial disclosure compliance is inherently complex, with over 39 registries, Most sponsors simply lack visibility into all the places they need to disclose information or what their commitments are. Automation can help make global compliance easier.

Beyond The Data Deluge: Immune Profiling In Early Immuno-Oncology Trials
This presentation explores how to design effective immune monitoring for early-phase immuno-oncology trials, focusing on strategic platform selection, cost, and operational feasibility.

How FAIR Data Principles Power Clinical Analytics Success
Explore key challenges in accessing and preparing analytics-ready clinical data, how FAIR guiding principles can help maximize the value of clinical trial data, and the impact of innovative analytics.

The Evolving Role Of AI And Innovation In Cardiac Monitoring
In this presentation, the panel of experts focuses on the changes AI has brought to cardiac monitoring data during clinical trials and addresses the skepticism over the use of new innovations.

Accelerating Data Cleaning
AI-enabled clinical trial technology enables faster processing of large data volumes, thereby enhancing workflows, easing the burden on busy trial teams, and protecting patients.

Streamline The Clinical Trial Disclosure Process
Core Data simplifies the registry submission and approval process by consolidating data for multiple registries into one single form, promoting consistency across registries and saving time by minimizing data entry.

Digging Through The Hype: Can AI/ML Assist Clinical Development?
AI has great potential to enhance the efforts of your team members by accelerating data interactions. Watch this webinar and discover how AI can improve clinical trial outcomes and data management.

The Importance Of Reliable Data To Clinical Trial Activation
Find an easier way to identify and enroll eligible patients for your clinical trials with quality data and site selection.

Importance And Requirements Of Study Results Posting
In addition to laws requiring disclosure of clinical trial results, the public also demands this information. TrialScope Chief Strategy Officer Thomas Wicks explains why sponsors should share study results.
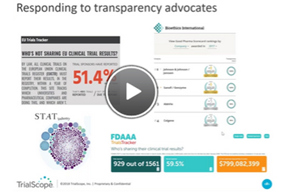
How To Boost Disclosure Efficiencies With Outsourcing
In this webinar, you’ll hear about exciting new advisory and managed disclosure services from TrialScope, including: compliance, policy and process assessments, as well as plain-language summaries, protocol registration, results posting, and redaction services.
...
The Cutting Edge Of Pharma: AI And RWD
Taken from a spotlight session at the CNS Summit, gain insights from thought leaders from Norstella and Citeline regarding artificial intelligence and real-world data.

How To Supercharge RWE Generation In Phase IV Trials
Watch as speakers discuss their joint efforts in implementing patient-centric digital trial solutions that harness the potential of RWD, translating it into actionable Real-World Evidence insights for a sponsoring client.

Citeline Study Feasibility
This predictive analytic solution helps sponsors select global study sites to successfully enroll more patients and accelerate clinical trial cycle times. Study Feasibility leverages both human expertise and machine-learning algorithms trained on Citeline’s best-in-...

Helping Sponsors Keep Up With The Latest Clinical Trials Intelligence
Trialtrove helps sponsors form development strategy, anticipate competitive activity, benchmark trial designs and analyze trends. Sponsors also enjoy direct access to industry-leading analysts.

Evolving Data Review With AI
AI is transforming clinical data review by automating processes and enhancing efficiency. Discover the four stages of evolution that emphasize automation, regulatory alignment, data analysis, and human oversight.

Verana Health: Where Quality Powers Insights
Drive innovation by improving your digital health in ophthalmology, urology, and neurology.

Pairing AI With High-Quality Data Leads To Clinical Success
While artificial intelligence (AI) is already impacting clinical trial planning, pairing AI with trusted data makes it even more powerful. Experts reveal how AI is being used today across feasibility, site selection, and trial strategy.

A Framework For Scalable Clinical Trial To Real World Data Linkage
Discover a scalable framework for data linkage in this on-demand webinar. Learn strategies to efficiently manage large datasets, enhance accuracy, and overcome common challenges in data integration.
