
TRIAL MANAGEMENT

Patient First Solutions For Oncology
Medable, Inc's new oncology offering is an end-to-end suite that includes an extensive eCOA library, pre-built and validated DCT applications such as Total Consent and Televisit, and protocol design consulting.
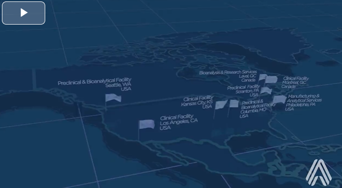
Altasciences Has You Covered From Coast To Coast
With your success in mind, we designed an integrated early phase drug development offering with eight locations, to get you from lead candidate selection to clinical proof of concept, and beyond.

Think U.S.A.: The Operations Behind Your Clinical Trials
Ready to think differently about early-phase clinical development? Check out the spotlight on our U.S. clinical facilities and the incredible teams behind them.

Why Disclosure As A Service?
Two clinical trial sponsors – one larger pharmaceutical company and one smaller biotech – share with TrialScope what their main reasons were for initially considering Disclosure as a Service. Watch the short video.

Are Your Clinical Study Meetings Building Momentum, Or Wasting Time?
Transforming passive clinical trial meetings into active catalysts for success, aligning teams and accelerating timelines.

Driving Simulation Studies At Altasciences
At Altasciences, we design driving studies to achieve your objectives, leveraging extensive experience across clinical testing of cognitive effects.

Yes, Your Study Is At Risk: Risk Mitigation Plans For Endpoints
Ocular endpoints often introduce unique bottlenecks that can stall enrollment and compromise data integrity. This presentation provides the tools for a streamlined, resilient clinical trial strategy.

Are Resource Constraints Impacting Your TMF Health?
Looking to quickly expand internal TMF resources with our anytime, anywhere TMF expertise? Unlock the potential to seamlessly access specialized TMF knowledge and support by viewing this presentation.

Keeping Up With ClinOps: Why And How To Improve Delivery
Industry experts discuss the concept of a trial platform as a service and how it can accelerate and enhance the day-to-day functions of clinical operations throughout the entire lifecycle of a study.

Practical Approaches To Faster Study Start-Ups
Learn how teams can leverage organizational, procedural, and technological solutions to recapture valuable days, weeks, and even months in the activation timelines of research sites.

TMF Management In Clinical Trials
Elevate your knowledge of clinical trials and the Trial Master File by exploring three critical topics.
Korio RTSM Platform: Real-Time Audit Trail Demo
Watch a real-time walkthrough of Korio’s built-in audit trail. Human-readable, blind-controlled, and fully exportable—see how sponsors get instant access to critical trial data.

Mastering Scope 3 Emissions In Clinical Trials
Explore innovative strategies for reducing Scope 3 emissions in clinical trials. Discover sustainable solutions that uphold quality and outcomes, crucial for achieving net-zero goals in healthcare research.
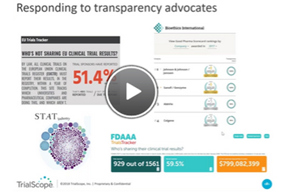
How To Boost Disclosure Efficiencies With Outsourcing
In this webinar, you’ll hear about exciting new advisory and managed disclosure services from TrialScope, including: compliance, policy and process assessments, as well as plain-language summaries, protocol registration, results posting, and redaction services.
...
The Real Payoff Of Reusable IRT/RTSM System Design
By utilizing consistent, repeatable system builds, sponsors gain significant advantages during protocol amendments and operational changes.

Disclosure As A Service ROI
How do sponsors determine the ROI of Disclosure as a Service? TrialScope talks with two sponsors – a large pharmaceutical company and a smaller biotech – for their take on this. Watch the short video.

Investigator SmartSelect
This advanced recommendation engine identifies protocol-specific investigators in just minutes, dramatically shortening a process that typically takes four to six weeks.

Say It With Technology: How Alnylam Fosters A Connected Relationship With Sites
Alnylam Pharmaceuticals is developing an entirely new class of medicines and delivered the world’s first and only approved RNAi therapeutics in 2018 and 2019. Learn how Alnylam earned its reputation among investigators as a sponsor of choice by standardizing studies...
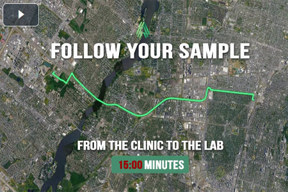
Accelerate Your Data With Our Co-Located Clinic And Lab
Watch as we take your fresh sample from our clinic to PBMC separation. This video overviews our optimized workflows that ensure timely processing, transport, and analysis of your samples.

Early Planning Makes Clinical Research Meetings More Effective
Starting earlier can dramatically improve clinical research meetings. Explore how timing affects your choices and why identifying non-negotiables first helps teams manage meeting budgets.

Clinical Trials And TMF 101 Learning Series: 2nd Session
The “Clinical Trials and TMF 101 Learning Series” kicks off it’s second session with three industry experts, that goes through some of the basics of clinical trials.

Evolving IRT For Today's Trials: Flexibility, Site Experience, And Integrated Workflow
Interactive Response Technology is evolving beyond traditional randomization and trial supply management to help guide protocol workflows in increasingly complex studies.
At The Crossroads Of Data: eCOA And Sensors Converging For Novel Insights
Leveraging sensors and eCOA addresses increasing clinical trial complexity and ensures a more efficient and effective study design.

Perceptive eClinical
Learn about a randomization and trial supply management (RTSM) solution that helps keep your trial on track.
Sponsors' Advice On Disclosure As A Service
Representatives from Teva and Lung Biotechnology share their insights for other sponsors considering Disclosure as a Service. Watch the short video now.

Tackling The EU CTR Compliance Challenge
Maintaining disclosure compliance is tricky enough with ClinicalTrials.gov, but when it comes to EU CTR compliance, the complexity rises to a whole new level. As a sponsor, do you find your organization “guilty” of these common errors?

Ensuring Compliance In Gene Therapy Trials: IRB And IBC Views
Explore ethical and regulatory challenges in neurological gene therapy trials, with expert insights on IRB/IBC collaboration, consent protocols, and oversight for vulnerable populations.
How RWD Reshapes Patient Recruitment Strategies
Discover how real-world data (RWD) is transforming patient recruitment by shifting from traditional site-centered methods to a patient-first approach.

Learning About The Sensors In myMedidata
Learn how sensor data and advanced analytics can transform clinical research and enable your study to overcome data challenges, improving patient care.

Going From Good To Great As A TMF Champion
Gain insight into regulatory nuances, best practices for documentation management, and strategies for fostering a culture of compliance within the team.
