Ignoring Digital Biomarkers Will Be Your Undoing

By Dan Schell, Chief Editor, Clinical Leader

Is the potential of digital biomarkers overblown? I mean, are they really going to be able to “revolutionize” clinical trials?
You know … I think they just might.
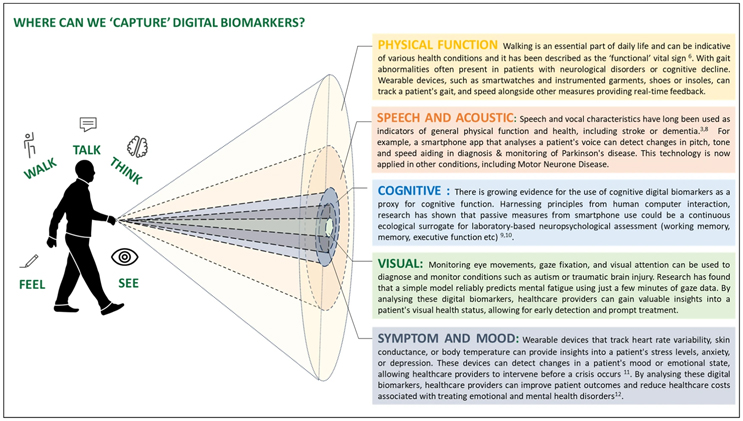
We’ve all seen the multitude of DHTs (digital health technologies) that are constantly popping up to enable some new digital biomarker. It seems these days, if you endeavor to gather additional data on some type of biomarker, there’s a device that can measure it for you — or, there’s tech in the works. I love this image from the article Walk, Talk, Think, See And Feel: Harnessing The Power Of Digital Biomarkers In Healthcare because it helps you visualize the diverse ways we can capture digital biomarkers.
WE NEED MORE DATA! NOW WHAT?
I knew Regeneron had developed a biomarker lab a few years ago, so I called long-time Clinical Leader editorial board member, Bari Kowal to learn more about how the company is viewing digital biomarkers. Not surprisingly, Kowal, who is SVP, development operations, portfolio management & biostatistics data management, was as optimistic as I was on the potential of this technology, but she was quick to note the challenges. “I think the field is still early in really understanding how to translate the data from some of these digital biomarker tools,” she says. “It’s difficult to figure out what you can measure repeatedly and with some sense of validity so you can conclude whatever difference you’ve come up with is due to the treatment. Of course, the other challenge is there’s so much data.”
Indeed, that continuous flow of real-time data creates a bit of a conundrum. After all, we have always wanted more ways to suss out some of the nuances related to how a trial participant is responding to an investigational drug outside of their regular clinic visits. Further, we’ve wanted that data to be as objective as possible. But now, with digital biomarkers, we could be receiving that exact type of data with every step or breath that patients take.
Kowal says that many of Regeneron’s research-related groups have been looking at computational science to try to change the way they invent molecules and utilize data for new insights. Consequently, in recent years, like many pharmas, they’ve found themselves seeking more data scientists or people with strong data backgrounds, as compared to traditional drug development expertise.
IT’S NOT JUST WEARBLES AND SENSORS
We’ve all heard of wearables, sensors, and remote-monitoring tools that are used to measure physiological function for patients with neuro disorders like Parkinson’s or Alzheimer’s. Vocal biomarkers also are used for studies analyzing depression, anxiety, and stress. Kowal mentioned sensors that help people with atopic dermatitis keep track of how many times they woke up in the night or how many times they scratched a part of their body.
In all these examples, some type of technology is providing objective data that can help support the clinical trial research; it’s not the treatment. It often eases the burden of participating in a trial by removing the need for patients to keep track of their symptoms or disease progression. For example, Kowal described a current Regeneron study that is targeting a pediatric population with an ultra-rare bone disease. Knowing that children, especially, could be fearful of participating in a clinical trial and reluctant to go to doctor appointments more frequently, Regeneron decided to gamify the patient diary and turn it into a video game. Patients can pick their avatar, and then the avatar goes through the process of being in the trial and gives the feedback. “At the end of it all, they can 3-D print their avatar and take it with them, which becomes a symbol of what they were able to accomplish,” explains Kowal. “So far, their responses have been fantastic. They were excited about doing something different — and fun — and doing something that they understood. And for us, it helped us gather the right data and retain patients who were interested in participating.”
EXPLORING THE CUTTING EDGE
Needless to say, Regeneron is all in on leveraging digital biomarkers in its current and upcoming trials across disease states. Like some of its competitors, the company hosts its own Digital Biomarker Summit (May 14, 2024) where experts from across the industry and academia convene to discuss the ongoing challenges, solutions, and advancements in this nascent, yet pivotal, space. In an upcoming article, I’ll talk with Regeneron’s Rinol Alaj, senior director, digital health technologies, about that event and the company’s digital biomarker lab.