It's A Fact: Sharing Clinical Trial Results With Participants Builds Trust
By Shalome Sine and Annick de Bruin, CISCRP

This article is part of a quarterly series highlighting key insights from the Perceptions and Insights Study, a global longitudinal survey study of over 12,800 patients and members of the public conducted in 2025. In this article, we will review what the survey results show about patient expectations for results‑sharing, actual rates of receiving study results, and the implications of this gap for trust and engagement in clinical research.
Clinical research participation can be logistically, emotionally, and physically demanding. Participants invest significant time and energy, often motivated by a desire to help advance science. Sharing study updates and results is a meaningful way to acknowledge their contribution, and patients expect basic communication about study progress and outcomes.
Evolving regulatory requirements and guidance also emphasize the importance of these transparency practices, including the European Union’s Clinical Trials Regulation, which mandates the return of trial results to participants and the public in plain language, as well as U.S. and U.K. requirements to inform patients and the public about study findings. Yet, despite regulatory and ethical direction, study results continue to be shared inconsistently, and progress toward routine implementation remains uneven across the industry.
This disconnect represents more than a missed communication opportunity. It has direct implications for participant satisfaction, patient/community engagement in drug development, and trust levels in pharmaceutical companies.
In this article, we explore findings from the Center for Information and Study on Clinical Research Participation’s (CISCRP) Perceptions & Insights Study, a global longitudinal survey conducted biennially since 2015, showing that patients consistently place a high value on receiving both individual-level results and overall study findings but only receive study reports/updates about half the time.
Patients Say Sharing Results Is “Very Important”
Since 2015, participants have consistently rated receiving study updates and results as a critical component of the trial experience. In the most recent 2025 survey, nearly nine in 10 respondents (88%) indicated that receiving regular study updates would be “somewhat” or “very important” to their participation experience.
Importantly, this sentiment is not limited to general updates during participation. When asked specifically about results after participation concludes, respondents express strong interest in both:
- individual study results and
- aggregate (overall) study findings.
Among those who received results, the perceived value is notable. In 2025, 94% of participants who received a summary of overall results described this information as “somewhat” or “very helpful.”
Patients Want Access To Their Individual Results
When patients report that they want their results returned, they are not asking for dense statistical tables or regulatory documents. Instead, CISCRP data show clear preferences for clinically relevant, understandable information.
In 2025, when asked what information they would be most interested in receiving after participation in a clinical trial, the top response was “my individual study results” (with 81% selecting). When asked which individual results they would be most interested in receiving, respondents most frequently selected:
- blood test results (85%)
- imaging results such as CT or PET scans (80%)
- genetic test results (78%)
- vital signs (e.g., blood pressure, heart rate) (70%)
- doctor visit notes (63%).
Among those who received individual study results (procedure/test results), well over half (59%) rated these results as “very helpful,” underscoring how personally meaningful this information can be.
Less Than Half Of Participants Receive End-Of-Trial Updates
Despite clear and consistent participant interest, actual rates of sponsors sharing results remain somewhat low.
Across the 2015–2025 period, fewer than half of clinical trial participants report receiving any reports or updates after completing a study. While there was a temporary increase during the COVID-19 pandemic, likely driven by heightened communication norms and digital adoption, rates have largely plateaued in recent years.
In 2025:
- 42% of participants reported receiving some form of study results after completion.
- 58% reported receiving none.
Most Respondents Indicate That Results Are Important, But Less Than Half Received Results After Participation
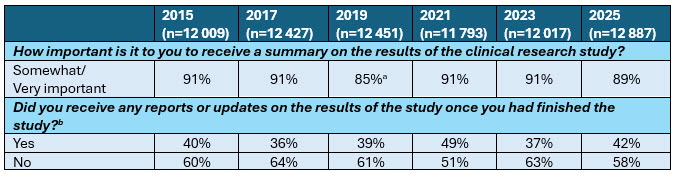
a This question was only asked among clinical trial participants in 2019. Percentage indicates the percentage of clinical trial participants in 2019 (n=3645).
b Base: Clinical trial participants. Excludes “I don’t remember.”
Notably, individual results were rated as more helpful and easier to understand than some higher-level scientific or regulatory updates, reinforcing the importance of tailoring communications to patient needs.
Differences By Experience, Therapeutic Area, And Demographics
CISCRP data also reveal meaningful subgroup differences that can help sponsors and sites prioritize and tailor their results-sharing strategies.
Participants with medical conditions in rare disease and oncology therapeutic areas were most interested in receiving their individual study results (with 93% and 88% indicating greatest interest in individual study results, respectively), likely reflecting the clinical relevance and emotional weight of test outcomes in these conditions.
Demographic patterns also emerge:
- Women are more likely than men to rate results-sharing as very important.
- Black/African American respondents consistently place higher importance on transparency-related practices, including receiving study updates and results.
These findings reinforce that results-sharing is not a nice-to-have for a narrow segment of participants; it is broadly valued, with particular importance among populations that clinical research is actively trying to engage and retain.
The Impact On Patient Engagement And Participation Experiences
Returning results is not only about closing the loop; it is closely tied to trust.
Across multiple waves of the Perceptions & Insights Study, respondents consistently identified transparency and information-sharing as key drivers of trust in pharmaceutical companies.
Among clinical trial participants, survey findings reflect the importance of sharing clinical trial results in building trust and patient engagement. Participants who received results were more likely to:
- report that the study “greatly exceeded” their expectations
- recommend participation in a clinical research study to family and friends
- place high trust in pharmaceutical companies.
Those Who Receive Study Updates Report Better Participation Experiences And Greater Trust In Pharma
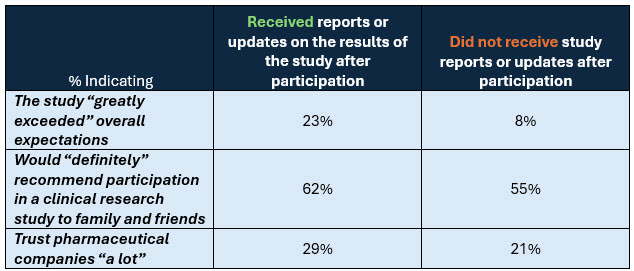
Base: Clinical trial participants. n=4,415
Source: CISCRP’s 2025 Perceptions and Insights Study
Conclusion
The data are clear: Returning study results aligns with participant expectations, enhances perceived value, and supports long-term engagement with clinical research.
As clinical research continues to emphasize patient engagement and diversity, returning results is one of the most tangible and measurable ways to demonstrate respect for participants as partners in the drug development process.
The CISCRP Perceptions & Insights Study consistently shows that clinical trial participants value results-sharing and respond positively when they receive this information. Yet the industry has made only incremental progress in delivering study results to patients after participation.
Maintaining communication with participants after study completion is a meaningful way to acknowledge their time, effort, and contributions to the drug development process. The Perceptions & Insights Study findings demonstrate that returning study results and sharing post-study updates are effective patient engagement practices, with measurable impacts on overall participation experiences, trust, and ongoing engagement with clinical research.
About The Authors:
 Annick de Bruin is the chief research and insights officer at the Center for Information and Study on Clinical Research Participation (CISCRP). She is responsible for the design, implementation, analysis, and reporting of a variety of CISCRP research studies, including the Perceptions & Insights studies and numerous patient advisory boards. She has more than 25 years of experience conducting primary and secondary research studies in the healthcare, life sciences, and consumer goods industries. She holds an MBA from the Graduate School of Management at Boston University and a Bachelor of Science degree from Bryant University.
Annick de Bruin is the chief research and insights officer at the Center for Information and Study on Clinical Research Participation (CISCRP). She is responsible for the design, implementation, analysis, and reporting of a variety of CISCRP research studies, including the Perceptions & Insights studies and numerous patient advisory boards. She has more than 25 years of experience conducting primary and secondary research studies in the healthcare, life sciences, and consumer goods industries. She holds an MBA from the Graduate School of Management at Boston University and a Bachelor of Science degree from Bryant University.
 Shalome Sine is a senior manager and quantitative insights specialist at the Center for Information and Study on Clinical Research Participation (CISCRP), where she leads efforts to elevate the patient voice in clinical research. Shalome believes that patient perspectives are essential to designing inclusive and efficient clinical trials. As part of CISCRP’s research services team, Shalome oversees both quantitative and qualitative patient voice initiatives, with a specialization in survey-based research that captures insights from patients and the public. She holds a Master of Public Health from Tufts University.
Shalome Sine is a senior manager and quantitative insights specialist at the Center for Information and Study on Clinical Research Participation (CISCRP), where she leads efforts to elevate the patient voice in clinical research. Shalome believes that patient perspectives are essential to designing inclusive and efficient clinical trials. As part of CISCRP’s research services team, Shalome oversees both quantitative and qualitative patient voice initiatives, with a specialization in survey-based research that captures insights from patients and the public. She holds a Master of Public Health from Tufts University.
