Wanted: More Women For Clinical Trials

By Dan Schell, Chief Editor, Clinical Leader

Diversity in clinical trials has been a recurring theme in my reporting during the past several years. Sponsors, CROs, and sites all acknowledge the importance of representative research populations, yet the industry continues to struggle with meaningful progress. In particular, the history of women participating in trials is, I’d say, concerning.
A new national survey conducted by UPMC’s Center for Connected Medicine and AMG Research takes a closer look at the issue. The report surveyed 400 women across the United States to better understand awareness of clinical trials, motivations for participation, and the barriers that prevent enrollment. The numbers illustrate the ongoing challenge. Women make up roughly 51% of the U.S. population but represent only about 41% of participants in clinical trials.
To delve deeper into this study and this topic, I spoke with Nicole Ansani, PharmD, SVP of new development initiatives at UPMC Enterprises and executive leader of UPMC Sponsored Clinical Trials and Partnerships (SCTP). Ansani spent more than two decades working on the pharmaceutical industry side before moving into her current role overseeing sponsored trials within the health system. That dual perspective gives her a unique vantage point on the structural challenges that affect recruitment. Her view is straightforward: Improving participation will require changes not only in how trials are communicated, but also in how they are designed and executed.
Awareness Exists — But Understanding Often Doesn’t
The survey findings suggest that most women are aware clinical trials exist, yet their understanding of what participation actually involves varies widely. Respondents were divided into three groups: women who had previously participated in a clinical trial, those who had considered participating but did not enroll, and those who had never considered participating at all.
Even among women who were generally aware of clinical trials, many reported limited knowledge about how studies work or what participation entails. That gap probably doesn’t surprise anyone who has worked in clinical research even for a short time. The unfortunate reality is that awareness alone does not necessarily translate into enrollment.
Ansani explained that women evaluate participation in trials through multiple lenses. “There’s the opportunity to contribute to the greater good, but there’s also the question of personal health benefits and practical considerations, such as financial compensation or reimbursement for time and travel.”
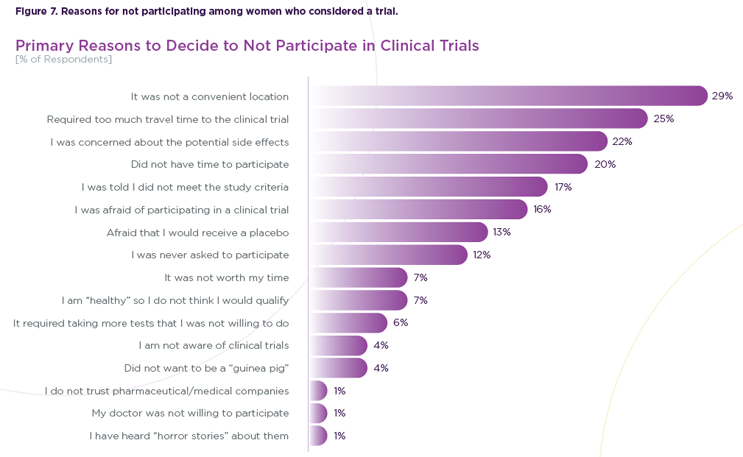
Fear And Logistics: Big Barriers
Among women who had not participated in clinical trials, fear of potential side effects emerged as the most common concern. Survey respondents who had never participated reported this concern at more than twice the rate of women who had previously taken part in a study. That difference suggests that uncertainty and perceived risk remain major barriers to first-time participation. The solution — or at least a solution — may seem simple: Rely on trusted clinicians to play a central role in overcoming the fears that anyone has regarding trial participation. “The data clearly showed that patients rely on their physicians as the most trusted source of information,” she said. “When a provider explains the risks and benefits, it makes a huge difference.”
But just having an awesome PI who can really alleviate any potential safety concerns isn’t going to solve the women enrollment problem. Probably (that’s my non-scientific disclaimer) more so than males, issues such as travel distance, visit frequency, and overall time commitment weigh heavy on the minds of female trial participants. Those constraints can quickly become deal breakers for patients managing careers, family responsibilities, and other daily obligations. Ansani pointed out that many women prioritize the health needs of family members before their own, which further complicates participation decisions. “Women often serve as the chief medical officer of their household,” she said. “They’re taking care of everyone else first. Their own health often comes last.”
Trial Design Still Plays A Role
Knowing Ansani had worked so long in Big Pharma, I couldn’t help but ask about the commonly heard complaint that protocols are too complex and maybe that’s why so many of these logistical barriers exist. She said she has a newfound perspective on this issue now that she’s on the site side. Namely, she says this “complex trial design” argument is really a shared responsibility between sponsors and sites. Sponsors ultimately design the protocol, but sites execute it. That means both sides influence how accessible a study becomes in practice. A protocol that requires frequent visits, complex procedures, or extensive travel will inevitably narrow the pool of participants able to enroll.
Ansani says, “It's really powerful when you can partner with pharma and ask questions such as:
- Are we hitting the mark with what you’re trying to solve for your clinical trial?
- Is that trial designed in a way that all the outcomes are necessary, useful, and important data points to ensure that the innovation makes it to market and can be used appropriately and effectively with our patients?
So, I see it as ‘joint solutioning;’ both of us trying to figure out how to use this propensity of clinical data to help design trials that maybe are more patient-friendly, that are less difficult from an outcomes perspective, and that enable more remote monitoring.” That last part is related to decentralization, and is key, considering for women balancing work and family responsibilities, even modest reductions in travel or visit burden could make participation more feasible.
Communication May Be The Simplest Fix
While protocol design often receives the most attention, Ansani believes some of the most practical improvements may come from better communication. Clinical trial documentation is notoriously complex. Informed consent forms can span dozens of pages, and even experienced clinicians sometimes struggle to summarize study details quickly during routine patient visits.
Ansani suggested that sponsors could make the process easier by providing plain-language summaries alongside clinical trial protocols. Similar summaries are already becoming common for published research articles and could help both clinicians and patients better understand the essentials of a study. “Imagine if every clinical trial protocol came with a plain-language overview,” she said. “Something clinicians could easily review and share with patients.” That type of resource could help physicians discuss trial options during routine care visits, which the survey suggests is the moment when many patients first learn about research opportunities. As mentioned earlier, healthcare professionals remain the most influential source of clinical trial information for women, far surpassing other communication channels.
For sponsors and sites alike, improving women’s participation will require more than awareness campaigns. It will require clearer communication, more practical trial designs, and a better understanding of how participation fits into patients’ daily lives. In many cases, the solutions are not particularly complex. They involve addressing everyday realities such as childcare, work schedules, travel time, and trust in healthcare providers. The challenge is ensuring those realities are considered early enough in trial planning to make a difference.
