
E-TECHNOLOGIES

Closing The "Compliance Gap" In TMF Archiving
Discover how organizations are crossing this “compliance gap” with PhlexTMF for Viewing, an easily accessible TMF archive for internal use, audits, and inspections.

Eliminating Custom Functions From EDC
Watch this video to see how companies like ALCON are driving faster study builds and higher quality for complex studies with data-driven rules and dynamics.

Qualified eSignature in Electronic Consent Systems
This on‑demand session offers a clear, practical overview of the major eSignature types used in eConsent — ranging from simple electronic signatures to fully qualified electronic signatures — and explains when each is appropriate.

Industry-First eTMF AI: PhlexTMF v21
Pre-trained on millions of documents and embedding the cumulative guidance of Phlexglobal’s TMF experts, uncover how teams can ensure clinical documents are “right-first-time” at document upload.

Building One Home for Sites
Watch this presentation and demonstration of One Home for Sites for the latest updates on how the industry is coming together — even competitors — to alleviate the technology burden on research sites.

The Role Of Technology In Improving Site Performance
Explore how clinical trial sponsors can benefit from the growing use of electronic source (eSource) technology at research sites.

An AI Action Plan For The TMF Professional
Explore AI's impact on TMF processes in this on-demand webinar. Gain clarity on AI's potential, key evaluation criteria, and practical applications for enhancing TMF health and inspection-readiness.

Central eSource: Leveraging Site Technology
In this presentation, CRIO founder and co-CEO Raymond Nomizu delves into how sponsors can leverage the electronic source trend among sites through the use of Central eSource.

Right-Size Your eConsent Approach
Delve into the art of tailoring the eConsent experience to match your study and patients' specific needs, such as crafting eConsent documents, selecting appropriate delivery models, and more.

Game On! Strategies For Conquering The Final Boss: TMF Close-Out
Much like playing a lengthy and challenging video game, managing the TMF from set-up through close-out can be an epic journey. Ever wondered how to ensure you're well-prepared to face the final boss?

Optimal eCOA For Vaccine Trials
Delve into the pivotal role that technologies like electronic Patient-Reported Outcomes (ePRO) can play in generating substantiating evidence and streamlining vaccine trial operations.

Fast, Flexible, And Proven Direct-From-Patient Data Capture With eCOA
Delve into how IQVIA eCOA captures patient data in a way that always keeps patients top of mind.

Rethinking Today's ePRO Delivery For Tomorrow's Clinical Trials
Willie Muehlhausen, Co-CEO and Founder of Safira Clinical Research, and Tim Davis, VP of Strategy at MyVeeva for Patients, discuss how ePRO delivery needs to change to better serve modern trials.

Selecting Medidata Rave: A Testimonial From Richmond Pharmacology
What made the Medidata's Rave EDC system stand out to Richmond Pharmacology during their vendor selection period?
Real-Time Data Tracking And Compliance Monitoring
Unlock real-time data tracking, customizable reporting, and proactive compliance monitoring with the IQVIA eCOA Sculptor Portal to enhance clinical decision-making and ensure protocol adherence.

Crossing The TMF Compliance Gap With Inspection-Ready Access For Closed Studies
Your drug study is closed, you need inspection-ready access to the Trial Master File, but your CRO is ready to get the TMF out of their system and wants to send you a zip folder full of documents. What do you do? In this session, we explore an innovative new way to ensure...

The Medable Platform: Deep Evidence Generation
Discover how Medable's platform technology enabled a top vaccine developer to accelerate study deployment and recruitment timelines.

Seamless And Integrated Onboarding For Trial Participants
Streamline your patient experience with a comprehensive, all-in-one solution that allows you to recruit, screen, and enroll participants remotely from the comfort of their own homes.

eTMF Innovation
Watch the available video to see how AI-Powered Automation can streamline your Trial Master File Management.
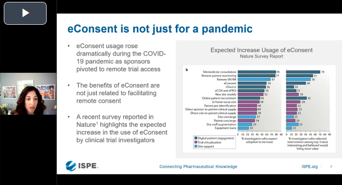
Agile eConsent: Taking A Flexible Approach To Global Implementation
Hear from industry experts on how they successfully combined agile eConsent technology with global regulatory expertise to increase enrollment rates and reduce both site and patient burden.

Electronic Data Capture Solutions For Your Phase I and Phase IV Studies
Take a cost-effective approach to meet your study's data capture, management, and analysis needs with a solution customized to your clinical trial.

Preparing For 2022
2022 will be an important year for drug development. There are many questions around changes that came into play during the pandemic. A panel of experts look back over the year and into the future to help you prepare for the coming year.

TMF Completeness: Simple Solutions For A Complex Problem
In this session, two of the top technology minds in TMF management will demonstrate and a simple yet innovative way to measure completeness at any point during the trial.
IQVIA eCOA Scribe Application
Experience seamless, secure, and flexible eCOA data collection with our application designed for diverse users, compatible with multiple devices, and equipped with offline functionality and robust authentication.
Overcoming TMF Management Challenges For Small And Mid-Sized Companies
Maintaining an inspection-ready Trial Master File is becoming increasingly burdensome, even for teams with significant resources. In this webinar our experts walk through a new, full-service TMF management approach implemented by smaller biopharmaceutical companies. By ad...
The Journey From File To Trial: Automating Regulatory To TMF
In this webinar, we explore the value of automation between the Trial Master File (TMF) and Regulatory solutions. This connection can bridge the gap from the clinical trial to the regulatory filing, ensuring consistency of information while reducing duplicated data ...

Combining De-Identified EHR And Claims Data
Learn how EHR+ Data can improve your understanding of the patient's journey in ophthalmology, urology, and neurology clinical trials.

Unleash The Power Of IQVIA eCOA: A Deep Dive
Edward Bartels, an expert in eCOA solutions, provides an in-depth overview of the market trends, emerging technologies, and how IQVIA eCOA is pioneering advancements in this space.

Elevating The Site Voice
A decentralized trial can be daunting for sites, but Medidata offers solutions to help site staff, sponsors, and patients move smoothly through the clinical trial process.
Auto Generate Documentation
Unlock seamless software development with IQVIA’s eCOA, which automates screenshots, validation documentation, and audit trails, ensuring real-time updates, easy exports, and full traceability.
