FEATURED ARTICLES
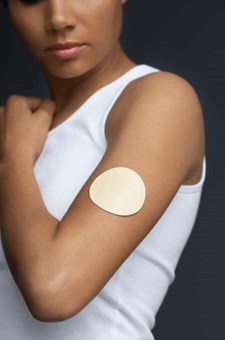 New Patch Technology Will Help Scintilla Treat Pain
New Patch Technology Will Help Scintilla Treat Pain
Pain management company Scintilla Pharmaceuticals has announced it will acquire SCILEX Pharmaceuticals to bolster its pain management business. SCILEX is engaged in the development and commercialization of products focused on the treatment of pain. Its lead product candidate, ZTlido, is a branded lidocaine patch formulation being developed for the treatment of postherpetic neuralgia, the chronic pain that sometimes develops with shingles.
-
Will Cyclobenzaprine Be A Solution For Fibromyalgia Patients?
Seth Lederman, M.D. has worked with many patients with fibromyalgia as a physician, and knows they have waited long enough for relief from its symptoms. “Those symptoms can quickly take over their lives,” he says. “Fibromyalgia is considered to be a pain disorder but it’s much more than that.”
-
Is There A Link Between The France Tragedy And Legal Highs?
By now you have heard about the tragic clinical trial in France that has resulted in the death of one person and the hospitalization of four others. While full details of what caused the fatal death of a healthy man are still not available, we do know the trial was likely testing an FAAH-inhibitor. Maia Szalavitz, writing on the website Vice.com, says this should be a wakeup call to some legal substance users in the U.S.
-
Your CRO Just Acquired Another CRO – Now What?
A year ago, Wendel Barr, CEO of CRO Synteract, was frustrated. His company was profitable, he had talented personnel, Synteract was managing a wide variety of projects, and his clients were satisfied with their level of service. Despite this success, his potential to grow the business was constrained. Synteract was a U.S.-based firm with limited presence overseas. As a result, his company often found itself forced to leave money on the table, especially as its customers moved to larger, Phase 3 trials. “It became clear to us that in order to provide the range of services our customers required, we had to have more offices in Europe, beyond our office in Prague,” he says. “If you want to be a full service provider from Phase 1 to Phase 4, you have to be global.”